Neonatal Seizure
 From Mdwiki
From Mdwiki | Neonatal seizure | |
|---|---|
| Other names: Seizures in neonates | |
 | |
| Specialty | Neurology, pediatrics |
| Symptoms | Seizure in newborn baby[1] |
| Complications | Brain injury[1] |
| Usual onset | Sudden, age 0-4 weeks[1] |
| Duration | Variable[1] |
| Causes | Reduced oxygen to the brain in babies born at term, bleeding into the brain in premature babies.[2] Other causes: cerebral infarction, infection, brain malformation, metabolic disorders.[2] |
| Risk factors | Preterm birth[2] |
| Diagnostic method | Electroencephalography (EEG).[2] |
| Treatment | Depends on cause[1] |
| Frequency | 1-3 per 1000 live births,[2] higher in preterm infants[3] |
A neonatal seizure is a seizure in a baby younger than age 4-weeks that is identifiable by an electrical recording of the brain.[1] Symptoms may be very subtle abnormal movements or none at all;[2] newborn babies have developmentally immature brains, and altered electrical activity presents differently to older age groups.[3] The seizures may vary from short recurrent ones to prolonged ones.[1]
Unlike in older children and adults, where most seizures are unprovoked, in young babies, seizures are typically a result of some degree of brain injury.[1] Commonest causes include reduced oxygen to the brain in babies born at term, and bleeding into the brain in premature babies.[2] Other causes include cerebral infarction, infection, brain malformation, or metabolic disorders.[2] There may be more than one reason for the occurrence of a neonatal seizure.[1] 5% to 10% of young babies with a seizure may be born with a neurological disease.[1]
Diagnosis is by electroencephalography (EEG).[2] Other tests include MRI, metabolic, and genetic testing.[2] When the first seizure occurs may indicate the cause.[1] Identifying the cause guides treatment.[1]
Neonatal seizures occur in around 1-3 of every 1000 live births.[2] It occurs more frequently in preterm babies.[1]
Classification[edit | edit source]
According to the International League against Epilepsy (ILAE), seizures are basically abnormally excessive or synchronous neuronal activity in the brain that is manifested as signs or symptoms. This definition, however, excludes the electrographic-only seizures that comprise 40-60% of that found in critically ill neonates[4]. As per the classification system by the American Clinical Neurophysiology Society, seizures can be classified into electroclinical, clinical only, and electrographic-only seizures[5].
Neonatal seizures have been classified into various types depending on various parameters. The earlier classification system, focused more on differentiating these neonatal seizures from those experienced in adult children. Neonates were found to experience either tonic or clonic seizures. They did not experience tonic-clonic seizures. If seizures were found to be focal, they were further classified into unifocal or multifocal.[6] Seizures in the neonatal population can be mainly categorized into acute symptomatic seizures and neonatal epilepsy that is related to genetic or structural factors.[7] Brain injury due to hypo-ischemic encephalopathy, ischemic stroke, intracranial hemorrhage or infection, inborn errors of metabolism, transient metabolic and brain malformations, lead to acute symptomatic seizures.[8] Neonatal epilepsy may be credited to genetic syndromes, developmental structural brain abnormalities, or metabolic diseases.[9] All the classification systems are found to be highly useful clinically.
Neonatal seizures are generally subclinical and their diagnosis based on the clinical observations is generally difficult.[10][11] Especially the seizures that involve senses are hard to detect in neonates. Diagnosis relies on identification of the cause of the seizure, and verification of actual seizure activity by measuring electrical activity with electroencephalography (EEG). The set of guidelines developed by the American Clinical Neurophysiology Society helps the healthcare providers know when the EEG is appropriate and corresponds to the seizures.[12] Treatment depends generally on the underlying cause of the seizure. Administration of deficient chemicals that lead to seizures treats it but if it still persists and is confirmed by EEG, pharmacologic treatment with anti-epileptic drugs is administered.
The considerable debate about the long-term consequence of a neonatal seizure exists between data and deductions reached through animal experimentations and those obtained through clinical investigations. The main conflicting issues are whether seizures in newborns can plant the roots for epileptogenesis and cause long-term deficits.[13] Fewer than half of the affected infants develop seizures in later life. Such neonatal seizures are considered self-limited, and thus the term neonatal epilepsy is not used to describe these seizures. It has been estimated that approximately 15% of neonatal seizures represent epilepsy syndrome.[14] The incidence of neonatal seizures has not been clearly established, although an estimated frequency of 80-120 cases per 100,000 neonates per year has been suggested. The incidence of seizures is higher in the neonatal period than at any other time of life,[15] and most often occurs in the first week of life.[16]
Signs and symptoms[edit | edit source]
These may be manifested in form of stiffening or jerking of limbs or trunk. Sometimes random eye movements, cycling movements of legs, tonic eyeball movements, and lip-smacking movements may be observed. Alteration in heart rate, blood pressure, respiration, salivation, pupillary dilation, and other associated paroxysmal changes in the autonomic nervous system of infants may be caused due to these seizures. Often these changes are observed along with the observance of other clinical symptoms.[17] A neonatal seizure may or may not be epileptic. Some of them may be provoked by stimulation or suppressed by restraining them.[17]
Seizures in the neonatal population often present differently than in other age groups due to brain immaturity.[18] Electroclinical seizures are defined by evidence of seizure activity on electroencephalogram as well as clinical signs or symptoms.[19] However, in neonates, there may be no clinical movement abnormality (either because the seizures are subclinical or because they were not witnessed), so the only symptom may be the abnormal level of consciousness. When motor movements do occur, they cannot be suppressed by gentle restraint by a nurse or caregiver.
Motor seizures[edit | edit source]
Classification systems have been developed based on neonatal seizure motor manifestations, summarized below.[20]
- Focal or multifocal clonic
Clonic seizures are defined by repetitive contractions of groups of muscles, typically of the limbs, face, or trunk.[21] These may involve one group of muscles (focal) or multiple groups of muscles (multifocal). An isolated focal seizure can move or spread, and can even alternate from one side of the body to the other. If they occur on both sides of the body, they may occur simultaneously in an asynchronous manner.[22] If a multifocal seizure, is limited to muscles of one side of the body, it may occur synchronously or asynchronously.[22] Focal clonic seizures cannot be suppressed by repositioning of limbs or by physical suppression.[23] Due to the neonatal brain's immaturity, the typical Jacksonian march may not occur. Focal seizures typically have very close correlates on EEG, with measurable EEG abnormalities with each seizure movement. The rhythm of the clonic movements and EEG abnormalities is usually slow, at 1-3 movements per second.
- Focal tonic
Focal tonic seizures are characterized by sustained muscle contraction of facial, limb, axial, and other muscle groups. It often involves asymmetric positioning of the neck and trunk and appears as abnormal posturing of a single limb. Horizontal eye deviation may or may not be involved.[23] They may be symmetric, asymmetric, focal, or multifocal. Such seizures cannot be provoked by stimulation or suppressed by restraint.
- Generalized tonic
A focal tonic seizure can generalize, or the first seizure can occur as a generalized seizure, or seizures that impair the neonate's level of consciousness. Generalized tonic seizures typically appear as symmetric and sustained posturing of limbs in either an extensor or flexor distribution. Generalized tonic seizures often manifest with the tonic extension of the upper and lower limbs and also may involve the axial musculature in an opisthotonic fashion. Generalized tonic seizures mimic decorticate posturing; the majority are not associated with electrographic seizures.Such seizures can be initiated by stimulation and can be suppressed by restraint.
- Myoclonic
Myoclonic movements can either be caused by seizures or be benign neonatal sleep myoclonus, a common mimicker of seizures in neonates. Myoclonic seizures are characterized by isolated and fast contractions of muscle groups that are non-repetitive. It generally involves flexor muscle groups of upper extremities- trunk, diaphragm, face.[24] These movements typically occur in the limbs or face. Stimulation can provoke myoclonic seizures.
- Spasms
Spasms include either flexor or extensor or both flexor and extensor. These occur in clusters and cannot be provoked by stimulation or suppressed by restraint.[17]
Subtle[edit | edit source]
Some clinicians use the term subtle seizures to describe seizures that appear to be more normal and there is an absence of distinct tonic or clonic movements but the presence of abnormal eye movements, stereotyped lip-smacking, or apneic events.[25] They are often overlooked and are generally assumed to be non-epileptic.[25] The random eye movement in them may be stimulated or increased by tactile stimulation while the progression movements such as bicycling, swimming, etc. can be restrained by repositioning.[22] Both the progression movements and oral-buccal lingual movements can be intensified and provoked upon stimulation.[22] Sometimes in subtle seizures complex limbic hyperactivity that is purposeless along with crying could be observed.[25]
Benign[edit | edit source]
Benign neonatal seizures are not classified as epilepsy.[26] A benign familial neonatal seizure onsets as early as 3 days of birth and may involve one or both sides of the brain.Recurrent seizure episodes are observed to occur in neonates.Electroencephalogram of infants with BFNS often have normal readings.Sometimes, they may show theta pointy, a specific abnormality. They usually begin with tonic stiffening accompanied by apnea.Later clonic jerks are witnessed.Electroencephalogram of infants with BFNS often have normal readings.Sometimes, they may show theta pointy, a specific abnormality.It occurs in 1 in every 1,00,000 newborns.[27]
This condition is usually inherited and is passed on in autosomal dominant manner. This condition is also caused due to mutation in KCNQ2 or KCNQ3 gene that may be carried by people bearing no family history of benign familial neonatal seizure. Mutation in these genes lead to excessive excitiability of neurons. Generally, patients with it are seen to bear mutation in KCNQ2 gene.[27]
Most of the infants with BFNS develop normally but some neonates with it may later develop intellectual disability which becomes evident in early childhood.In some patients approximately 15%, epilepsy occurs once in life after seizures associated with BFNS are gone away. Myokymia is also witnessed in a few cases.[27]
Pathophysiology[edit | edit source]
During the neonatal developmental stages, numerous pathophysiological mechanisms, lead to excessive excitation and reduced inhibition, which lowers their seizure threshold when compared with that of adults. This has been proved in animal(rodent) models wherein the adult and infant models are administered with the chemoconvulsant agent and their threshold seizure potential is compared.[28] This lowered seizure threshold potential makes the neonatal brain susceptible to acute symptomatic seizures.
Decreased inhibition[edit | edit source]
Gamma-butyric acid (GABA) is the main inhibitory neurotransmitter in adult humans. Upon binding with its receptor i.e. GABAa, it causes hyperpolarisation of the neuronal membrane by causing the net influx of chloride ions. This hyperpolarisation leads to inhibition of further action potentials. However, in neonates, there is a relatively high expression of NKCC1(sodium-potassium-chloride cotransporter 1) than KCC2(potassium-chloride cotransporter 2). NKCC1 causes net efflux of chloride ions[29] while KCC2 is responsible for causing a net influx of chloride ions. Increased expression of NKCC1 leads to depolarisation of the neuronal membrane. This depolarisation removes voltage-dependent Mg from N-methyl-D-aspartate(NMDA) receptors and triggers calcium influx. The binding of calcium to the receptors causes the generation of secondary messengers that increases the risks of seizures and increase the excitability of the brain.[30][31]
Increased activation[edit | edit source]
Glutamate is the primary excitatory neurotransmitter and the expression of its receptor is developmentally regulated.[32] It binds to NMDA receptors, kainite receptors, and AMPA receptors. In course of the developmental stages, in several parts of the brain, a subunit of NMDA receptor-GluN2B is highly expressed which increases calcium influx. This mechanism increases the duration of postsynaptic currents in the immature brain in comparison to adult brains.[33]
Causes[edit | edit source]
Neonatal seizures have a number of causes. Determining the cause of a confirmed seizure is important because treatment and prognosis vary based on underlying etiology of the seizure. In contrast to seizures that occur in other age groups, seizures that occur during the neonatal period are most often caused by the following processes:
- Hypoxic-Ischemic Encephalopathy: This is the most common cause of seizures in the neonatal period[34] and is an underlying cause for 40-45% of neonatal seizures. The onset of seizure associated with it occurs within first 12 to 24 hrs of life. In HIE, identification of occurrence of seizure is important as it occurs in approximately 25-50℅ of people with this condition. If it's not diagnosed it may further increase the brain injury. Seizure semiology and severity varies with the location and size of injured region of the brain. Use of EEG becomes extremely necessary for seizure cases that are subclinical.
- Perinatal Arterial Stroke: Arterial stroke can be caused by intra-arterial thrombosis or embolism from the heart or placenta. The risk for perinatal arterial stroke increases with a variety of conditions that occur due to material factors during birth (oligohydramnios, chorioamnionitis, placental abnormalities) or neonatal factors (clotting disorders, congenital heart defects,[34] coagulation problems, systemic infection, male sex, placental abnormalities). The onset of seizures associated with focal strokes begin after 24hrs to 48 hrs of birth. Focal clonic seizure is generally associated with it due to involvement of motor cortex in middle cerebral artery region.
- Intraventricular hemorrhage: It causes generally causes seizures in term infants except large hemorrhage involving injury to parenchyma.
- Central Nervous System Infection: CNS Infection are found in 3-10% of neonates who seize, though exact incidence varies between studies.[34] Bacterial meningitis and viral meningoencephalitis are most prevalent, though fungal infections can occur in antenatal, perinatal and prenatal brain of neonates. Infections caused due to late streptococcus B and herpes simplex virus often occur at any time during the neonatal period. Lumbar puncture help in diagnosis of neonates suspected of it. In case of the infant is unstable for it, empirical treatment of meningoencephalitis is warranted. Seizures related to it persist longer than those associated with HIE or ICH.
- Congenital central nervous system malformations: Lissencephaly, polymicrogyria, and tuberous sclerosis are specific entities known to cause seizures due to defects in brain tissue development. Since prognosis is poor and often these disorders are genetic, identification of this etiology is of utmost importance to be able to counsel parents appropriately.
- Inborn errors of metabolism: Inborn errors of metabolism can cause physiologic conditions that result in seizures. These errors are genetic and often are accompanied by other symptoms such as lethargy, poor feeding, and low tone.[34] Diagnosis often involves specific laboratory tests of metabolic products as well as genetic tests. Several classification systems exist for seizures caused by inborn errors of metabolism, one of which separates causes into problems with neurotransmitter metabolism, energy production, and biosynthetic substances crucial for brain formation.[35]
- Electrolyte abnormalities: Metabolic abnormalities such as hypoglycemia, hyponatremia, and hypocalcemia can manifest as seizures.
- Substance-Related: Neonatal abstinence syndrome occurs when maternal drug use before birth results in a fetal withdrawal syndrome. Substances include alcohol, cocaine, narcotics, tricyclclic antidepressants, or other sedatives. Seizures can be prevented from occurring if the symptoms of withdrawal are recognized and treated early.[34]
Diagnosis[edit | edit source]
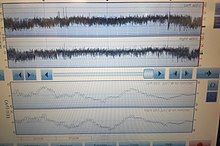
Seizure activity in a neonate is difficult to diagnose, as many seizures have no clinical correlate. Altered level of consciousness is often the only clue, and in a neonate this can be difficult to accurately assess. Thus, diagnosis relies on attempting to directly measurement the abnormal electrical activity in the brain with electroencephalography (EEG). EEG is combined with video recording of the infant to correlate any seizure movements with EEG recordings.
There are several modes of EEG that are commonly used in the NICU to diagnose neonatal seizures. Conventional continuous multichannel conventional EEG is the gold standard for diagnosis of epileptiform activity, but requires expert interpretation. Newer amplitude integrated EEG (aEEG) (also termed cerebral function monitoring, or CFM) allows easier monitoring of brain activity, but may not allow identification of short duration, low amplitude, or very high frequency seizure activity.[36] Often, both modes are displayed concurrently.
Determining the cause[edit | edit source]
Since many causes of seizures can be rapidly reversed and longterm sequelae prevented, evaluation of underlying cause is of utmost importance. Evaluation for infection (with blood counts, lumbar puncture, and empiric treatment with antibiotics) often occurs during EEG monitoring. Blood glucose and electrolyte testing can identify metabolic problems that can be corrected. Further testing includes evaluation for genetic causes and other more rare metabolic causes.[37] Brain injury such as cerebral infarction or hemorrhage can be evaluated with imaging techniques such as magnetic resonance imaging (MRI) and brain ultrasound to avoid unnecessary radiation exposure.
Differential diagnosis[edit | edit source]
Infants can exhibit stereotyped movements that may be hard to distinguish from seizure activity. Since many of these non-seizure movements are not dangerous and require no treatment, differentiation from actual seizure activity is useful. Jitteriness is common in the neonatal period and is seen in upwards of 2/3 of neonates. It is characterized by a tremor that is especially prominent during sleep or periods of agitation. Gaze deviation or eye movements do not occur. Benign neonatal sleep myoclonus (BNSM) is another common movement that can be mistaken for a seizure. It is characterized by jerking limb movements only during sleep, and stop with waking of the infant. BNSM typically occurs later in infancy, but can occur during the neonatal period.
Treatment[edit | edit source]
Once diagnosis is made, the goals of management are to identify the cause of the seizure, stop the seizure activity, and maintain physiologic parameters such as oxygenation, ventilation, blood glucose, and temperature.
Treatment greatly depends on the cause of the seizure. For example, infectious causes of seizures (meningitis, meningoencephalitis), are often treated with antimicrobials (antibiotic, antifungal, or antiviral medications). Similarly, electrolyte or glucose abnormalities are treated by repleting or lowering the offending electrolyte or sugar.
If the cause of the seizures are unlikely to be easily or quickly corrected, once diagnosis of a seizure is made, the mainstay of treatment is pharmacotherapy with anti-epileptic drugs. A 2013 systematic review found that most practitioners use phenobarbital or phenytoin.[38] This study found that phenobarbital has the safest side effect profile and longest history of use in neonates.Hence, phenobarbital is used for first-line of treatment. After just one dose of 20 mg/kg of it, seizures are in control, in approximately half of the patients diagnosed with acute symptomatic seizures.[39] A NEOLEV2 study was conducted in the year 2020 to compare the efficiency of levetiracetam and phenobarbital as the first line of therapy. 80% of patients who were administered phenobarbital became seizure-free in 24hrs whereas merely 28% patients became seziure free in case of levetiracetam administration. Benzodiazepines are often used as second-line treatment if treatment with phenobarbital does not result in clinical improvement. Almost 66% of patients with acute symptomatic seizures, don't have a complete response to the initial dose administered. This is valid for any type of medication selected.[40] Research is ongoing on the use of other anti-epileptics that are commonly used in older children and adults and are safe or efficacious to use in neonates. Part of the challenge of anticonvulsant drug treatment during the neonatal period is that the immature excitatory and inhibitory neurotransmitter system results in few effective drug targets.[37] There are some drugs that are effective in controlling seizures. "Phenobarbitone remains the first-line treatment for seizures in most countries throughout the world, though off-label use of newer anticonvulsants is widespread in the USA[41]”. The United States prefers to use unprescribed drugs to help slow down neonatal seizures, but they also use prescribed drugs if needed. Drugs are the only option infants are given, even though they are not 100% effective, they are the best option in helping take control of the seizures to stop them from occurring.
The following table lists the dosage of the respective medicine:[42]
| Medicine | Dode |
|---|---|
| Phenobarbitone | Target level: 40mcg/mL
Maintenance dose: 3-6mg/kg/d Loading dose: Intravenously 20mg/kg, repeat once when required |
| Phenytoin | Target level: 10-20mcg/ml to be administered over ten minutes
Maintenance dose: 2.5-5 mg/kg/d to be administered in two equal doses. Loading dose: 20mg/kg phenytoin equivalent intravenous over 20 minutes |
| Levetiracetam | Target level: Unknown
Maintenance dose: 30-60mg/kg/d in three equal doses Loading dose: Intravenously 40-60mg/kg/d |
Outcomes[edit | edit source]
With earlier diagnosis and increasingly prompt diagnosis, mortality after diagnosis of neonatal seizures has decreased dramatically from an estimated 33% in the 1990s to around 10% in the 2010s.[43] Underlying cause of the seizure remains the greatest predictor of ongoing seizures and neurologic problems later in life. Controversy remains with the extent of damage the seizures themselves cause. Clinician consensus is that frequent or intractable seizures (status epilepticus) leads to neuronal damage and are associated with later neurodevelopment problems.[44]
Developmental delay, cerebral palsy, and problems with vision and hearing are the most common poor outcomes following neonatal seizures.[43] Severity of impairment ranges greatly and many infants develop normally once the initial seizure cause is treated.
Studies have identified risk factors for poor outcomes after neonatal seizures. Infants that are premature, have hypoxemic ischemic encephalopathy, CNS infection, severe intraventricular hemorrhage, structural central nervous system defect, or severely abnormal EEG tracings tend to do worse than infants with focal strokes, transient metabolic issues (hypoglycemia, hypocalcemia), or clinical seizures without EEG abnormalities.[43] There are many other risk factors for neonatal seizures. "Some of the risk factors that we have detected, such as low Apgar score, need for resuscitation at birth, and perinatal distress, were previously reported[45]”. An apgar score indicates how a baby is immediately after birth. Perinatal distress is when a mother experiences a lot of stress during pregnancy. This shows how serious neonatal seizures really are, and not many people knew that.
Risk of epilepsy later during childhood is a common question asked by parents in the neonatal intensive care unit (NICU). Unfortunately, the answer is rarely clear cut as it depends greatly on the underlying cause. For all infants with neonatal seizures regardless of cause, the rate of subsequent seizures during childhood is estimated between 10 and 20%.[46] Infants who survive severe global HIE have the highest rate of epilepsy later in life.[43]
Epidemiology[edit | edit source]
It is difficult to determine the incidence of seizures in the neonatal period. Estimations range between 1-5 per 1,000 live births,[37] though the actual rate of seizures during this period may be higher due to a lack of accurate diagnosis of sub-clinical seizure activity without continuous EEG monitoring. The epidemiology moreover varies in high-income countries (HIC) from that in low-income ones (LIC). The incidence is estimated to be 1-3 per 1000 live births in HIC whereas it ranges from 36-90 per live birth in LIC. Acute causes of seizures (hypoxemic ischemic encephalopathy, infection, intracranial hemorrhage, stroke, etc.) are more common than the first episode of neonatal epilepsy syndromes. Watching the EEG monitor and detecting when a seizure occurs can help the preterm infant survive these seizures. "Continuous monitoring and cranial imaging in the setting of neonatal seizures now compromise the standard of care at many centers, and these technologies are being used to guide management, diagnosis, and prognosis[47]". As the monitoring devices for neonatal seizures become more advanced, the closer researchers are to finding how these seizures occur and how they can be stopped.
Seizures occur during the neonatal period at a higher rate than in any other period of life. Seizures in the developing brain are more common than in a mature brain for several reasons. First, the developing brain is hyperexcitable due to excess in excitatory glutaminergic neurons and immaturity of inhibitory gamma-amino butyric acid (GABA) neurons. Preterm infants are at especially high risk for seizures for this reason.[43]
The incidence of seizures is more common in the neonatal stage than in other stages of life.[48] Neonatal seizures are comparatively rare and affect 1 or 3.5 in 1000 infants born.[49] They are the most frequent neurological problem in the nursery that is associated with greater risks of morbidity and mortality,[50][51] often requiring evaluation and treatment in a neonatal intensive care unit. Better care delivered in neonatal care units, with improved healthcare facilities, has decreased the mortality rate associated with these seizures. However, the long-term morbidity rate remains approximately the same.[52] The increased risk of neonatal seizures may be attributed to risks related to delivery, gestation, post-natal period, and physiological factors.
Research directions[edit | edit source]
Since interpretation of continuous EEG monitoring requires a trained neurologist, automated interpretation software has been proposed. Algorithms and machine learning have been studied, however logistical and mathematic challenges remain.
See also[edit | edit source]
References[edit | edit source]
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 Garfinkle, Jarred; Miller, Stephen P. (2022). "110. Neonatal seizures". In Jankovic, Joseph; Mazziotta, John C.; Pomeroy, Scott L. (eds.). Bradley and Daroff's Neurology in Clinical Practice. Vol. II. Neurological disorders and their management (8th ed.). Edinburgh: Elsevier. pp. 2022–2039. ISBN 978-0-323-64261-3. Archived from the original on 2022-06-10. Retrieved 2022-06-02.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Ramantani, Georgia; Schmitt, Bernhard; Plecko, Barbara; Pressler, Ronit M.; Wohlrab, Gabriele; Klebermass-Schrehof, Katrin; Hagmann, Cornelia; Pisani, Francesco; Boylan, Geraldine B. (October 2019). "Neonatal Seizures-Are We there Yet?". Neuropediatrics. 50 (5): 280–293. doi:10.1055/s-0039-1693149. ISSN 1439-1899. PMID 31340400. Archived from the original on 2022-05-01. Retrieved 2022-06-03.
- ↑ 3.0 3.1 Krawiec, Conrad; Muzio, Maria Rosaria (2022). "Neonatal Seizure". StatPearls. StatPearls Publishing. Archived from the original on 2022-04-03. Retrieved 2022-06-02.
- ↑ Mizrahi, E. M.; Kellaway, P. (1987-12-01). "Characterization and classification of neonatal seizures". Neurology. 37 (12): 1837–1837. doi:10.1212/wnl.37.12.1837. ISSN 0028-3878. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Tsuchida, Tammy N.; Wusthoff, Courtney J.; Shellhaas, Renée A.; Abend, Nicholas S.; Hahn, Cecil D.; Sullivan, Joseph E.; Nguyen, Sylvie; Weinstein, Steven; Scher, Mark S.; Riviello, James J.; Clancy, Robert R. (2013-04). "American Clinical Neurophysiology Society Standardized EEG Terminology and Categorization for the Description of Continuous EEG Monitoring in Neonates". Journal of Clinical Neurophysiology. 30 (2): 161–173. doi:10.1097/wnp.0b013e3182872b24. ISSN 0736-0258. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
{{cite journal}}: Check date values in:|date=(help) - ↑ Rose, Arthur L.; Lombroso, Cesare T. (1970-03-01). "NEONATAL SEIZURE STATES : A Study of Clinical, Pathological, and Electroencephalographic Features in 137 Full-term Babies with a Long-term Follow-up". Pediatrics. 45 (3): 404–425. doi:10.1542/peds.45.3.404. ISSN 0031-4005. S2CID 245089207. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ "Recent Advances in the Diagnosis and Treatment of Neonatal Seizures - Yahoo Search Results". search.yahoo.com. Archived from the original on 2022-06-10. Retrieved 2022-01-30.
- ↑ Beleza, Pedro (May 2012). "Acute symptomatic seizures: a clinically oriented review". The Neurologist. 18 (3): 109–119. doi:10.1097/NRL.0b013e318251e6c3. ISSN 2331-2637. PMID 22549348. S2CID 25710798. Archived from the original on 2022-01-30. Retrieved 2022-05-27.
- ↑ Axeen, Erika J. T.; Olson, Heather E. (2018-06-01). "Neonatal epilepsy genetics". Seminars in Fetal and Neonatal Medicine. NEONATAL SEIZURE. 23 (3): 197–203. doi:10.1016/j.siny.2018.01.003. ISSN 1744-165X. PMID 29426807. S2CID 46818810. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Glass, Hannah C.; Wusthoff, Courtney J.; Shellhaas, Renée A.; Tsuchida, Tammy N.; Bonifacio, Sonia Lomeli; Cordeiro, Malaika; Sullivan, Joseph; Abend, Nicholas S.; Chang, Taeun (2014-04-08). "Risk factors for EEG seizures in neonates treated with hypothermia: a multicenter cohort study". Neurology. 82 (14): 1239–1244. doi:10.1212/WNL.0000000000000282. ISSN 1526-632X. PMC 4001204. PMID 24610326.
- ↑ Clancy, Robert R.; Legido, Agustin; Lewis, Donald (1988). "Occult Neonatal Seizures". Epilepsia. 29 (3): 256–261. doi:10.1111/j.1528-1157.1988.tb03715.x. ISSN 1528-1167. PMID 3371282. S2CID 24190612. Archived from the original on 2022-02-02. Retrieved 2022-05-27.
- ↑ Shellhaas, Renée A.; Chang, Taeun; Tsuchida, Tammy; Scher, Mark S.; Riviello, James J.; Abend, Nicholas S.; Nguyen, Sylvie; Wusthoff, Courtney J.; Clancy, Robert R. (December 2011). "The American Clinical Neurophysiology Society's Guideline on Continuous Electroencephalography Monitoring in Neonates". Journal of Clinical Neurophysiology. 28 (6): 611–617. doi:10.1097/WNP.0b013e31823e96d7. ISSN 0736-0258. PMID 22146359. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Lombroso CT (2007). "Neonatal seizures: gaps between the laboratory and the clinic". Epilepsia. 48 Suppl 2: 83–106. doi:10.1111/j.1528-1167.2007.01070.x. PMID 17571356.
- ↑ Shellhaas RA, Wusthoff CJ, Tsuchida TN, Glass HC, Chu CJ, Massey SL, Soul JS, Wiwattanadittakun N, Abend NS, Cilio MR (August 2017). "Profile of neonatal epilepsies: Characteristics of a prospective US cohort". Neurology. 89 (9): 893–899. doi:10.1212/WNL.0000000000004284. PMC 5577964. PMID 28733343.
- ↑ Sheth RD, Hobbs GR, Mullett M (January 1999). "Neonatal seizures: incidence, onset, and etiology by gestational age". Journal of Perinatology. 19 (1): 40–3. doi:10.1038/sj.jp.7200107. PMID 10685200.
- ↑ Plu, Fred (1983). "Status epilepticus. Mechanisms of brain damage and treatment". Annals of Neurology. 34 (6): 1–551. doi:10.1002/ana.410150628. PMID 6829325.
- ↑ 17.0 17.1 17.2 MD, Blaise F. Bourgeois; MD, Edwin Dodson; MD, Douglas R. Nordli, Jr; MD, John M. Pellock; PhD, Raman Sankar, MD (2007-12-16). Pediatric Epilepsy: Diagnosis and Therapy. Demos Medical Publishing. ISBN 978-1-934559-86-4. Archived from the original on 2022-05-23. Retrieved 2022-05-27.
- ↑ Al-Fraik, Nasren Gamal Saleh; Al-Khurum, Khadeejah Miftah Ali (2020-09-27). "Neonatal Seizure in Tobruck Medical Center". Scientific Journal of Applied Sciences of Sabratha University. 3 (2): 13–24. doi:10.47891/sabujas.v3i2.13-24. ISSN 2708-7301. S2CID 229010547. Archived from the original on 2022-02-05. Retrieved 2022-05-27.
- ↑ Pressler, Ronit M.; Cilio, Maria Roberta; Mizrahi, Eli M.; Moshé, Solomon L.; Nunes, Magda L.; Plouin, Perrine; Vanhatalo, Sampsa; Yozawitz, Elissa; Vries, Linda S. de; Vinayan, Kollencheri Puthenveettil; Triki, Chahnez C. (2021). "The ILAE classification of seizures and the epilepsies: Modification for seizures in the neonate. Position paper by the ILAE Task Force on Neonatal Seizures". Epilepsia. 62 (3): 615–628. doi:10.1111/epi.16815. ISSN 1528-1167. PMID 33522601. S2CID 231762346. Archived from the original on 2022-02-05. Retrieved 2022-05-27.
- ↑ Scher, Mark S. (December 1998). "Diagnosis and management of neonatal seizures.By Eli M. Mizrahi and Peter Kellaway Philadelphia, Lippincott-Raven, 1998 192 pp, illustrated, $79.00". Annals of Neurology. 44 (6): 988. doi:10.1002/ana.410440628. ISSN 0364-5134.
- ↑ Abend, Nicholas S.; Jensen, Frances E.; Inder, Terrie E.; Volpe, Joseph J. (2018-01-01), Volpe, Joseph J.; Inder, Terrie E.; Darras, Basil T.; de Vries, Linda S. (eds.), "Chapter 12 - Neonatal Seizures", Volpe's Neurology of the Newborn (Sixth Edition), Elsevier, pp. 275–321.e14, doi:10.1016/b978-0-323-42876-7.00012-0, ISBN 978-0-323-42876-7, archived from the original on 2022-02-06, retrieved 2022-02-06
- ↑ 22.0 22.1 22.2 22.3 MD, Blaise F. Bourgeois; MD, Edwin Dodson; MD, Douglas R. Nordli, Jr; MD, John M. Pellock; PhD, Raman Sankar, MD (2007-12-16). Pediatric Epilepsy: Diagnosis and Therapy. Demos Medical Publishing. ISBN 978-1-934559-86-4. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ 23.0 23.1 Volpe, Joseph J. (1989-09-01). "Neonatal Seizures: Current Concepts and Revised Classification". Pediatrics. 84 (3): 422–428. doi:10.1542/peds.84.3.422. ISSN 0031-4005. PMID 2671912. S2CID 39194127. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Krawiec, Conrad; Muzio, Maria Rosaria (2022), "Neonatal Seizure", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 32119422, archived from the original on 2022-02-28, retrieved 2022-02-06
- ↑ 25.0 25.1 25.2 Panayiotopoulos, C. P. (2005). Neonatal Seizures and Neonatal Syndromes. Bladon Medical Publishing. Archived from the original on 2022-05-12. Retrieved 2022-05-27.
- ↑ National Institute for Health and Clinical Excellence (January 2012). "9" (PDF). The Epilepsies: The diagnosis and management of the epilepsies in adults and children in primary and secondary care. National Clinical Guideline Centre. pp. 119–129. Archived (PDF) from the original on 2013-12-16. Retrieved 2022-05-27.
- ↑ 27.0 27.1 27.2 "Benign familial neonatal seizures: MedlinePlus Genetics". medlineplus.gov. Archived from the original on 2022-02-13. Retrieved 2022-02-13.
- ↑ Stafstrom, Carl E.; Thompson, James L.; Holmes, Gregory L. (February 1992). "Kainic acid seizures in the developing brain: status epilepticus and spontaneous recurrent seizures". Developmental Brain Research. 65 (2): 227–236. doi:10.1016/0165-3806(92)90184-x. ISSN 0165-3806. PMID 1572066. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Dzhala, Volodymyr I.; Talos, Delia M.; Sdrulla, Dan A.; Brumback, Audrey C.; Mathews, Gregory C.; Benke, Timothy A.; Delpire, Eric; Jensen, Frances E.; Staley, Kevin J. (November 2005). "NKCC1 transporter facilitates seizures in the developing brain". Nature Medicine. 11 (11): 1205–1213. doi:10.1038/nm1301. ISSN 1078-8956. PMID 16227993. S2CID 25348736. Archived from the original on 2022-03-09. Retrieved 2022-05-27.
- ↑ Nardou, Romain; Ferrari, Diana C.; Ben-Ari, Yehezkel (2013-08-01). "Mechanisms and effects of seizures in the immature brain". Seminars in Fetal and Neonatal Medicine. Neonatal seizures. 18 (4): 175–184. doi:10.1016/j.siny.2013.02.003. ISSN 1744-165X. PMID 23702158. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Rivera, C.; Voipio, J.; Payne, J. A.; Ruusuvuori, E.; Lahtinen, H.; Lamsa, K.; Pirvola, U.; Saarma, M.; Kaila, K. (1999-01-21). "The K+/Cl- co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation". Nature. 397 (6716): 251–255. Bibcode:1999Natur.397..251R. doi:10.1038/16697. ISSN 0028-0836. PMID 9930699. S2CID 7687596. Archived from the original on 2022-04-20. Retrieved 2022-05-27.
- ↑ Monyer, Hannah; Burnashev, Nail; Laurie, David J.; Sakmann, Bert; Seeburg, Peter H. (March 1994). "Developmental and regional expression in the rat brain and functional properties of four NMDA receptors". Neuron. 12 (3): 529–540. doi:10.1016/0896-6273(94)90210-0. ISSN 0896-6273. PMID 7512349. S2CID 26733598. Archived (PDF) from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Carrasco, Melisa; Stafstrom, Carl E. (2018). "How Early Can a Seizure Happen? Pathophysiological Considerations of Extremely Premature Infant Brain Development". Developmental Neuroscience. 40 (5–6): 417–436. doi:10.1159/000497471. ISSN 1421-9859. PMID 30947192. S2CID 96432850. Archived from the original on 2022-01-16. Retrieved 2022-05-27.
- ↑ 34.0 34.1 34.2 34.3 34.4 Vasudevan C, Levene M (August 2013). "Epidemiology and aetiology of neonatal seizures". Seminars in Fetal & Neonatal Medicine. 18 (4): 185–91. doi:10.1016/j.siny.2013.05.008. PMID 23746578.
- ↑ Van Hove JL, Lohr NJ (November 2011). "Metabolic and monogenic causes of seizures in neonates and young infants". Molecular Genetics and Metabolism. 104 (3): 214–30. doi:10.1016/j.ymgme.2011.04.020. PMID 21839663.
- ↑ Boylan GB, Stevenson NJ, Vanhatalo S (August 2013). "Monitoring neonatal seizures". Seminars in Fetal & Neonatal Medicine. 18 (4): 202–8. doi:10.1016/j.siny.2013.04.004. PMID 23707519.
- ↑ 37.0 37.1 37.2 Glass HC (March 2014). "Neonatal seizures: advances in mechanisms and management". Clinics in Perinatology. 41 (1): 177–90. doi:10.1016/j.clp.2013.10.004. PMC 3925308. PMID 24524454.
- ↑ Slaughter LA, Patel AD, Slaughter JL (March 2013). "Pharmacological treatment of neonatal seizures: a systematic review". Journal of Child Neurology. 28 (3): 351–64. doi:10.1177/0883073812470734. PMC 3805825. PMID 23318696.
- ↑ Painter, Michael J.; Scher, Mark S.; Stein, Aryeh D.; Armatti, Stacey; Wang, Zhiming; Gardiner, Joseph C.; Paneth, Nigel; Minnigh, Beth; Alvin, John (1999-08-12). "Phenobarbital Compared with Phenytoin for the Treatment of Neonatal Seizures". New England Journal of Medicine. 341 (7): 485–489. doi:10.1056/NEJM199908123410704. ISSN 0028-4793. PMID 10441604. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Ziobro, Julie; Shellhaas, Renée A. (April 2020). "Neonatal Seizures: Diagnosis, Etiologies, and Management". Seminars in Neurology. 40 (2): 246–256. doi:10.1055/s-0040-1702943. ISSN 0271-8235. Archived from the original on 2022-06-10. Retrieved 2022-06-10.
- ↑ Boylan, Geraldine B.; Stevenson, N. J.; Vanhatalo, S. (August 2013). "Elsevier Enhanced Reader". Seminars in Fetal and Neonatal Medicine. Neonatal seizures. 18 (4): 202–208. doi:10.1016/j.siny.2013.04.004. PMID 23707519. Archived from the original on 2021-09-21. Retrieved 2021-09-21.
- ↑ Vegda, Hemadri; Krishnan, Vaisakh; Variane, Gabriel; Bagayi, Vaishnavi; Ivain, Phoebe; Pressler, Ronit M. (2022-01-20). "Neonatal Seizures—Perspective in Low-and Middle-Income Countries". Indian Journal of Pediatrics. 89 (3): 245–253. doi:10.1007/s12098-021-04039-2. ISSN 0019-5456. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ 43.0 43.1 43.2 43.3 43.4 Uria-Avellanal C, Marlow N, Rennie JM (August 2013). "Outcome following neonatal seizures". Seminars in Fetal & Neonatal Medicine. 18 (4): 224–32. doi:10.1016/j.siny.2013.01.002. PMID 23466296.
- ↑ Clancy RR, Legido A (February 1991). "Postnatal epilepsy after EEG-confirmed neonatal seizures". Epilepsia. 32 (1): 69–76. doi:10.1111/j.1528-1157.1991.tb05614.x. PMID 1985832. S2CID 25884790.
- ↑ Pisani, Francesco; Facini, C.; Bianchi, E.; Giussani, G.; Piccolo, B.; Beghi, E. (June 2020). "Elsevier Enhanced Reader". Epilepsy & Behavior. 107: 107075. doi:10.1016/j.yebeh.2020.107075. PMID 32304988. S2CID 215767632. Archived from the original on 2022-06-10. Retrieved 2021-09-21.
- ↑ Tekgul H, Gauvreau K, Soul J, Murphy L, Robertson R, Stewart J, Volpe J, Bourgeois B, du Plessis AJ (April 2006). "The current etiologic profile and neurodevelopmental outcome of seizures in term newborn infants". Pediatrics. 117 (4): 1270–80. doi:10.1542/peds.2005-1178. PMID 16585324. S2CID 31487112.
- ↑ Glass, Hannah C.; Kan, J.; Bonifacio, S. L.; Ferriero, D. M. (February 2012). "Elsevier Enhanced Reader". Pediatric Neurology. 46 (2): 111–115. doi:10.1016/j.pediatrneurol.2011.11.006. PMC 3266555. PMID 22264706.
- ↑ Volpe, Joseph J. (2008-06-11). Neurology of the Newborn E-Book. Elsevier Health Sciences. ISBN 978-1-4377-2079-2. Archived from the original on 2022-05-23. Retrieved 2022-05-27.
- ↑ Vasudevan, Chakrapani; Levene, Malcolm (August 2013). "Epidemiology and aetiology of neonatal seizures". Seminars in Fetal and Neonatal Medicine. 18 (4): 185–191. doi:10.1016/j.siny.2013.05.008. ISSN 1744-165X. PMID 23746578. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
- ↑ Pisani, Francesco; Cerminara, Caterina; Fusco, Carlo; Sisti, Lisa (2007-12-04). "Neonatal status epilepticus vs recurrent neonatal seizures: Clinical findings and outcome". Neurology. 69 (23): 2177–2185. doi:10.1212/01.wnl.0000295674.34193.9e. ISSN 0028-3878. PMID 18056582. S2CID 39133720. Archived from the original on 2022-03-15. Retrieved 2022-05-27.
- ↑ Pisani, Francesco; Barilli, Angela Luciana; Sisti, Lisa; Bevilacqua, Giulio; Seri, Stefano (2008-01-01). "Preterm infants with video-EEG confirmed seizures: Outcome at 30 months of age". Brain and Development. 30 (1): 20–30. doi:10.1016/j.braindev.2007.05.003. ISSN 0387-7604. PMID 17964748. S2CID 25487534. Archived from the original on 2022-06-11. Retrieved 2022-05-27.
- ↑ Ramantani, Georgia (December 2013). "Neonatal epilepsy and underlying aetiology: to what extent do seizures and EEG abnormalities influence outcome?". Epileptic Disorders. 15 (4): 365–375. doi:10.1684/epd.2013.0619. ISSN 1294-9361. PMID 24342861. S2CID 22568118. Archived from the original on 2022-06-10. Retrieved 2022-05-27.
Categories: [Epilepsy] [Symptoms and signs] [Neonatology]
↧ Download as ZWI file | Last modified: 05/15/2024 15:39:17 | 4 views
☰ Source: https://mdwiki.org/wiki/Neonatal_seizure | License: CC BY-SA 3.0
 KSF
KSF