Igg4-Related Disease
 From Mdwiki
From Mdwiki | IgG4-related disease | |
|---|---|
| Other names: IgG4-related systemic disease | |
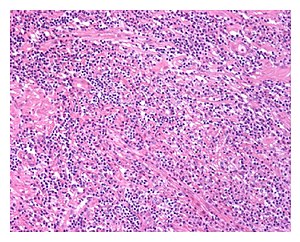 | |
| Low power view of IgG4-related prostatitis. The prostatic stroma shows a dense inflammatory infiltrate and fibrosis (H&E, 100x) | |
| Specialty | Immunology, rheumatology |
IgG4-related disease (IgG4-RD), formerly known as IgG4-related systemic disease, is a chronic inflammatory condition characterized by tissue infiltration with lymphocytes and IgG4-secreting plasma cells, various degrees of fibrosis (scarring) and a usually prompt response to oral steroids. In approximately 51–70% of people with this disease, serum IgG4 concentrations are elevated during an acute phase.[1][2][3]
It is a relapsing–remitting disease associated with a tendency to mass forming, tissue-destructive lesions in multiple sites, with a characteristic histopathological appearance in whichever site is involved. Inflammation and the deposition of connective tissue in affected anatomical sites can lead to organ dysfunction, organ failure, or even death if not treated.[4]
Early detection is important to avoid organ damage and potentially serious complications.[5] Treatment is recommended in all symptomatic cases and also in asymptomatic cases involving certain anatomical sites.[4][6]
Signs and symptoms[edit | edit source]
IgG4-related disease has been described as an indolent condition. Although possibly based on opinion rather than on objective assessments, symptoms, if any, are commonly described as mild in the medical literature.[citation needed] This can be in spite of considerable underlying organ destruction. People are often described as being generally well at the time of diagnosis, although some may give a history of weight loss.
Pain is generally not a feature of the inflammation. However it may occur as a secondary effect, for example due to either obstruction or compression.
Often diagnosis is made due to the presence of painless swellings or mass lesions, or due to complications of masses, e.g. jaundice due to involvement of the pancreas, biliary tree or liver. Symptoms are commonly attributed to other conditions and other diagnoses may have been made years before diagnosis, e.g. urinary symptoms in men attributed to common prostate conditions. Lesions may also be detected incidentally on radiological images, but can be easily misdiagnosed as malignancies.[citation needed]
Reported cases do include some significant symptoms or findings however:
| System | Uncommon symptoms and complications |
|---|---|
| Neurological | Seizures,[7][8] paralysis or hemiparesis,[7] cranial nerve palsies,[7] sensorineural hearing loss,[9] pituitary hormone deficiencies[10] |
| Eye | Loss of vision,[7] proptosis[11] |
| Cardiovascular | Constrictive pericarditis,[12] heart block,[13] ruptured aortic aneurysm,[14][15] aortic dissection,[16] carotid artery dissection,[17] intracranial aneurysm,[18] angina,[19] sudden cardiac death[20][21] |
| Respiratory | Airway obstruction,[22] pleural effusion[23][24] |
| Gastrointestinal | Esophageal obstruction,[25][26] bowel obstruction[27] |
| Urological | Renal failure,[28] hydronephrosis,[28][29] testicular pain[30] |
Organ manifestations[edit | edit source]
IgG4-RD can involve one or multiple sites in the body. With multiorgan involvement, the sites involved can be affected at the same time (synchronously) or at different unrelated periods (metachronously).
Several different diseases that have been known for many years are now considered to be manifestations of IgG4-RD. These include: type 1 autoimmune pancreatitis, interstitial nephritis, Riedel's thyroiditis, Mikulicz's disease, Küttner's tumor, inflammatory pseudotumors (in various sites of the body), mediastinal fibrosis and some cases of retroperitoneal fibrosis.[31][32]
| Organ or site | Preferred names | Previously used names |
|---|---|---|
| Head and neck | ||
| Salivary gland | IgG4-related sialadenitis:
|
Mikulicz's disease (salivary and lacrimal glands),[34] chronic sclerosing sialadenitis, Küttner's tumour (submandibular glands) |
| Orbit | IgG4-related ophthalmic disease (IgG4-ROD) including:
|
Mikulicz's disease (salivary and lacrimal glands),[34] Idiopathic orbital inflammatory disease, orbital pseudotumor |
| Paranasal sinuses[35] | Chronic sinusitis, Eosinophilic angiocentric fibrosis (upper respiratory tract and orbit)[36] | |
| Pharynx | IgG4-related pharyngitis[37] | |
| Thyroid gland | IgG4-related thyroid disease | Riedel's thyroiditis, Riedel's struma |
| Soft tissues of the head and neck | Idiopathic cervical fibrosis,[38] sclerosing cervicitis, cervical fibrosclerosis | |
| Central Nervous System | ||
| Pituitary gland | IgG4-related hypophysitis:
|
Autoimmune hypophysitis |
| Meninges | IgG4-related pachymeningitis (dura mater), IgG4-related leptomeningitis[39][40] (arachnoid and pia mater) |
Idiopathic hypertrophic pachymeningitis |
| Chest and abdomen | ||
| Pancreas | IgG4-related pancreatitis | Type 1 autoimmune pancreatitis, lymphoplasmacytic sclerosing pancreatitis, 'chronic pancreatitis with diffuse irregular narrowing of the main pancreatic duct'[41] |
| Lung | IgG4-related lung disease | Pulmonary inflammatory pseudotumour |
| Pleura | IgG4-related pleuritis | |
| Liver | IgG4-related hepatopathy | |
| Bile duct | IgG4-related sclerosing cholangitis | |
| Gallbladder | IgG4-related cholecystitis | |
| Aorta (especially the infrarenal portion) |
IgG4-related aortitis, IgG4-related periaortitis[42] |
Inflammatory aortic aneurysm, Chronic sclerosing aortitis, chronic periaortitis. |
| Branches of the aorta (including coronary,[35] renal or iliac arteries) |
IgG4-related periarteritis[42] | |
| Pericardium | IgG4-related pericarditis | |
| Mediastinum | IgG4-related mediastinitis | Fibrosing mediastinitis, chronic sclerosing mediastinitis |
| Retroperitoneum | IgG4-related retroperitoneal fibrosis | Retroperitoneal fibrosis, Albarran-Ormond syndrome, Ormond's disease, perirenal fasciitis, Gerota's fasciitis/syndrome, periureteritis fibrosa, sclerosing lipogranuloma, sclerosing retroperitoneal granuloma, non-specific retroperitoneal inflammation, sclerosing retroperitonitis, retroperitoneal vasculitis with perivascular fibrosis.[43] |
| Mesentery | IgG4-related mesenteritis (subtypes are: mesenteric panniculitis, mesenteric lipodystrophy and retractile mesenteritis)[43] | Sclerosing mesenteritis, systemic nodular panniculitis, liposclerosis mesenteritis, mesenteric Weber–Christian disease, mesenteric lipogranuloma, xanthogranulomatous mesenteritis.[43] |
| Breast | IgG4-related mastitis | Sclerosing mastitis |
| Genitourinary | ||
| Kidney | IgG4-related kidney disease (IgG4-RKD):
|
Idiopathic tubulointerstitial nephritis |
| Prostate | IgG4-related prostatitis | |
| Vas deferens | IgG4-related perivasal fibrosis[30][44] | Chronic orchialgia |
| Scrotum | IgG4-related paratesticular pseudotumor,[33][45] IgG4-related epididymo-orchitis[33] |
Paratesticular fibrous pseudotumor, inflammatory pseudotumor of the spermatic cord, pseudosarcomatous myofibroblastic proliferations of the spermatic cord, proliferative funiculitis, chronic proliferative periorchitis, fibromatous periorchitis, nodular periorchitis, reactive periorchitis, fibrous mesothelioma[46] |
| Other | ||
| Lymph nodes | IgG4-related lymphadenopathy | |
| Skin | IgG4-related skin disease | Angiolymphoid hyperplasia with eosinophilia,[47] cutaneous pseudolymphoma[48] |
| Nerve | IgG4-related perineural disease[33][49] | |
This is not a complete list, as IgG4-RD can involve any site in the body.
Other affected sites, confirmed on histology to be manifestations of IgG4-RD, include: heart;[13] hard palate,[50] esophagus,[25][26] stomach,[51] small intestine,[52] rectum,[53] adrenal gland,[54] ovary,[55] uterus,[23] ureter,[56] bladder,[57] urachus,[58] and synovium.[59] Approximately 1/3 of cases exhibit increases in blood eosinophil counts, either eosinophilia or hypereosinophilia.[citation needed]
Radiologic evidence suggestive of involvement of the superior vena cava[13] and seminal vesicle[60] has been reported in confirmed cases of IgG4-RD.
Diagnosis[edit | edit source]
Diagnosis requires tissue biopsy of an affected organ with characteristic histological findings. Serum immunoglobulin G4 is often elevated but this is not always the case.[citation needed]
Histology[edit | edit source]
Whatever area of the body is involved, the hallmark histopathological features of IgG4-RD are:[3][31][32]
- A dense lymphoplasmacytic (lymphocytes and plasma cells) infiltrate rich in IgG4-positive plasma cells.
- IgG4 immunostaining needs to be specifically requested and performed in order to detect IgG4-positive plasma cells.
- Fibrosis, arranged at least focally[3] in a "storiform" pattern.
- "Storiform" is commonly referred to as meaning 'having a cartwheel pattern', but its literal meaning is the appearance of 'a woven mat [Latin: storea] (of rush or straw)'.
- Obliterative phlebitis.
Other histopathological features associated with IgG4-RD are:
Submandibular gland[edit | edit source]
In an article from 1977, histological research into 349 cases of Küttner's tumor (now known as 'IgG4-related sialadenitis') identified four distinct stages of the fibroinflammatory process:[61]
- Stage 1: Focal periductal (around the salivary ducts) infiltration of lymphocytes
- Stage 2: Diffuse infiltration of lymphocytes and severe periductal fibrosis (scarring around the salivary ducts)
- Stage 3: Prominent infiltration of lymphocytes, atrophy of parenchyma (i.e. loss of functional areas due to shrinkage), and periductal sclerosis (scarring resulting in hardening around the salivary ducts)
- Stage 4: Marked loss of and sclerosis (hardening) of the parenchyma (functional area) - similar to the process involved in cirrhosis where there is shrinkage and loss of functional areas of the liver
This may reflect the inflammatory process and development of fibrosis that occurs in other organs involved in IgG4-RD.
Treatment[edit | edit source]
The goal of treatment is the induction and maintenance of remission so as to prevent progression of fibrosis and organ destruction in affected organ(s).
An international panel of experts have developed recommendations for the management of IgG4-RD.[4][6] They concluded that in all cases of symptomatic, active IgG4-RD that treatment is required. Some cases with asymptomatic IgG4-RD also require treatment, as some organs tend to not cause symptoms until the late stages of disease. Urgent treatment is advised with certain organ manifestations, such as aortitis, retroperitoneal fibrosis, proximal biliary strictures, tubulointerstitial nephritis, pachymeningitis, pancreatic enlargement and pericarditis.
Induction of remission[edit | edit source]
In untreated patients with active disease, the recommended first-line agent for induction of remission is glucocorticoids unless contraindications exist. Glucocorticoids characteristically result in a rapid and often dramatic improvement in clinical features and often a resolution of radiographic features. However, where advanced fibrotic lesions have resulted in irreversible damage, the response to glucocorticoids and other current treatment options may be poor or even absent.
Although not validated yet in clinical trials, the common induction regime is prednisolone 30–40 mg per day for 2–4 weeks, then gradually tapered over 3 to 6 months. Recurrences during or after tapering of glucocorticoids are frequent however. Steroid-sparing immunosuppressive agents might be considered, depending on local availability of these drugs, for use in combination with glucocorticoids from the start of treatment in order to reduce the side-effects of prolonged glucocorticoid usage. Steroid-sparing agents that have been used include rituximab, azathioprine, methotrexate, and cyclophosphamide, although trials are needed to ascertain the effectiveness of each drug in IgG4-RD.
Maintenance[edit | edit source]
Following a successful induction of remission, maintenance therapy might be given in some cases, for example when there is a high risk of relapse or in patients with organ-threatening manifestations. Common maintenancy therapy is prednisolone 2.5–5 mg per day, or use of a steroid-sparing agent instead.
Relapse[edit | edit source]
Relapses are common, and a previous history of relapse appears to be a strong predictor of future relapse. When relapse occurs while off therapy and there has been a prolonged disease remission following initial glucocorticoid induction, then the relapse can usually be managed successfully with a re-induction strategy using glucocorticoids. Introducing a steroid-sparing agent might also need to be considered for relapses; however, none has been tested in prospective, controlled studies, and evidence for their efficacy beyond that offered by concomitant glucocorticoid therapy is scarce.[5]
In one retrospective cohort study, baseline concentrations of serum IgG4, IgE and blood eosinophils were found to be independently predictive of relapse risk following treatment with rituximab with or without glucocorticoids; the higher the baseline values, the greater the relapse risk and the shorter the time to relapse.[62]
Other interventions[edit | edit source]
When organ involvement causes local mechanical problems, further organ-specific interventions may be necessary. For example, when a tumefactive lesion causes obstruction of the bile duct, it may be necessary to insert a biliary stent to allow the bile to drain freely.
Similarly, ureteral or vascular stents, surgical resection or radiotherapy may be considered for various different presenting problems.
Trials[edit | edit source]
Research is also under way to evaluate the effect and safety of plasmablast-directed therapy with a monoclonal antibody (XmAb5871) which inhibits B-cell function without depleting these immune cells.[63][64] XmAb5871 targets CD19 with its variable domain and has an Fc domain that has increased affinity to FcγRIIb.[65]
Epidemiology[edit | edit source]
As recognition of IgG4-RD is relatively recent, there are limited studies on its epidemiology. It is therefore difficult to make an accurate estimation of prevalence. Furthermore, age of onset is almost impossible to estimate; age at diagnosis is frequently misused as the age of onset.
A 2011 study estimated the incidence of IgG4-RD in Japan at 2.8–10.8/million population, with a median age of onset of 58 years.[66]
Nomenclature[edit | edit source]
| Previously names |
|---|
IgG4-related systemic disease (IgG4-RSD)
IgG4-related sclerosing disease IgG4-related systemic sclerosing disease IgG4-related autoimmune disease IgG4-associated multifocal systemic fibrosis IgG4-associated disease IgG4 syndrome Hyper-IgG4 disease Systemic IgG4-related plasmacytic syndrome IgG4-positive multiorgan lymphoproliferative syndrome |
Prior to 2011, IgG4-RD used to get mentioned in the medical literature under various different names.[2][66]
At the 2011 International Symposium on IgG4-Related Diseases Archived 2017-02-23 at the Wayback Machine, the consensus name of IgG4-related disease was endorsed for the condition.[2] This name had already been agreed upon as a consensus name among Japanese investigators,[2][3] notably choosing not to use the term 'systemic' as that might lead to malignant tumours in other organs getting incorrectly diagnosed as being just another manifestation of the IgG4-related condition.[66]
However, some experts at the international symposium did express reservations about naming the disease after IgG4, as its role in pathogenesis is questionable and the use of serum IgG4 concentrations as a biomarker is unreliable.[2]
An expanded term, 'Immunoglobulin G4-related disease', has sometimes been used also.[67] However, this term was never referenced in the 2012 recommendations for nomenclature,[2] and its use would appear to be erroneous.
See also[edit | edit source]
References[edit | edit source]
- ↑ Wallace, Zachary S.; Deshpande, Vikram; Mattoo, Hamid; Mahajan, Vinay S.; Kulikova, Maria; Pillai, Shiv; Stone, John H. (26 August 2015). "IgG4-Related Disease: Clinical and Laboratory Features in One Hundred Twenty-Five Patients". Arthritis & Rheumatology. 67 (9): 2466–2475. doi:10.1002/art.39205. PMC 4621270. PMID 25988916.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 John H. Stone; Arezou Khosroshahi; Vikram Deshpande; John K. C. Chan; J. Godfrey Heathcote; Rob Aalberse; Atsushi Azumi; Donald B. Bloch; William R. Brugge; Mollie N. Carruthers; Wah Cheuk; Lynn Cornell; Carlos Fernandez-Del Castillo; Judith A. Ferry; David Forcione; Günter Klöppe; Daniel L. Hamilos; Terumi Kamisawa; Satomi Kasashima; Shigeyuki Kawa; Mitsuhiro Kawano; Yasufumi Masaki; Kenji Notohara; Kazuichi Okazaki; Ji Kon Ryu; Takako Saeki; Dushyant Sahani; Yasuharu Sato; Thomas Smyrk; James R. Stone; Masayuki Takahira; Hisanori Umehara; George Webster; Motohisa Yamamoto; Eunhee Yi; Tadashi Yoshino; Giuseppe Zamboni; Yoh Zen; Suresh Chari (October 2012). "Recommendations for the nomenclature of IgG4-related disease and its individual organ system manifestations". Arthritis & Rheumatism. 64 (10): 3061–3067. doi:10.1002/art.34593. PMC 5963880. PMID 22736240.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Vikram Deshpande; Yoh Zen; John KC Chan; Eunhee E Yi; Yasuharu Sato; Tadashi Yoshino; Günter Klöppe; J Godfrey Heathcote; Arezou Khosroshahi; Judith A Ferry; Rob C Aalberse; Donald B Bloch; William R Brugge; Adrian C Bateman; Mollie N Carruthers; Suresh T Chari; Wah Cheuk; Lynn D Cornell; Carlos Fernandez-Del Castillo; David G Forcione; Daniel L Hamilos; Terumi Kamisawa; Satomi Kasashima; Shigeyuki Kawa; Mitsuhiro Kawano; Gregory Y Lauwers; Yasufumi Masaki; Yasuni Nakanuma; Kenji Notohara; Kazuichi Okazaki; Ji Kon Ryu; Takako Saeki; Dushyant V Sahani; Thomas C Smyrk; James R Stone; Masayuki Takahira; George J Webster; Motohisa Yamamoto; Giuseppe Zamboni; Hisanori Umehara; John H Stone (18 May 2012). "Consensus statement on the pathology of IgG4-related disease". Modern Pathology. 25 (9): 1181–1192. doi:10.1038/modpathol.2012.72. PMID 22596100. S2CID 7677776. Archived from the original on 21 September 2021. Retrieved 7 May 2021.
- ↑ 4.0 4.1 4.2 Arezou Khosroshahi; Zachary S. Wallace; Jayne Littlejohn Crowe; Takashi Akamizu; Atsushi Azumi; Mollie N. Carruthers; Suresh T. Chari; Emanuel Della-Torre; Luca Frulloni; Hiroshi Goto; Phillip A. Hart; Terumi Kamisawa; Shigeyuki Kawa; Mitsuhiro Kawano; Myung-Hwan Kim; Yuzo Kodama; Kensuke Kubota; Markus M Lerch; Matthias Löhr; Yasufumi Masaki; Shoko Matsui; Tsuneyo Mimori; Seiji Nakamura; Takahiro Nakazawa; Hirotaka Ohara; Kazuichi Okazaki; Jay H. Ryu; Takako Saeki; Nicolas Schleinitz; Akira Shimatsu; Tooru Shimosegawa; Hiroki Takahashi; Masayuki Takahira; Atsushi Tanaka; Mark Topazian; Hisanori Umehara; George J. Webster; Thomas E. Witzig; Motohisa Yamamoto; Wen Zhang; Tsutomu Chiba; John H. Stone (July 2015). "International Consensus Guidance Statement on the Management and Treatment of IgG4-Related Disease". Arthritis & Rheumatology. 67 (7): 1688–1699. doi:10.1002/art.39132. PMID 25809420. S2CID 39750214. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ 5.0 5.1 Terumi Kamisawa; Yoh Zen; Shiv Pillai; John H. Stone (11 April 2015). "IgG4-related disease". The Lancet. 385 (9976): 1460–1471. doi:10.1016/S0140-6736(14)60720-0. PMID 25481618. S2CID 28886899. Archived from the original on 18 February 2017. Retrieved 7 May 2021.
- ↑ 6.0 6.1 Walsh, Nancy (1 April 2015). "Experts Weigh In on IgG4 Disease - Diagnosis requires biopsy, and treatment starts with steroids". MedPage Today. Archived from the original on 3 March 2016. Retrieved 6 March 2016.
- ↑ 7.0 7.1 7.2 7.3 Somalinga Nagendran Karthik; K. Bhanu; Sakthi Velayutham; M. Jawahar (July–September 2011). "Hypertrophic pachymeningitis". Annals of Indian Academy of Neurology. 14 (3): 203–204. doi:10.4103/0972-2327.85896. PMC 3200046. PMID 22028536.
- ↑ Young-Sub Lee; Hye Won Lee; Ki-Su Park; Seong-Hyun Park; Jeong-Hyun Hwang (October 2014). "Immunoglobulin G4-Related Hypertrophic Pachymeningitis with Skull Involvement". Brain Tumor Research and Treatment. 2 (2): 87–91. doi:10.14791/btrt.2014.2.2.87. PMC 4231624. PMID 25408931. Archived from the original on 2016-03-05. Retrieved 2021-05-07.
- ↑ Jesica Romina Gallo; Alberto Christian Ortiz; Sergio Oscar Paira (September 2014). "IgG4-Related Disease and Sensorineural Hearing Loss". Clinical and Experimental Otorhinolaryngology. 7 (3): 236–237. doi:10.3342/ceo.2014.7.3.236. PMC 4135164. PMID 25177444.
- ↑ Akira Shimatsu; Yutaka Oki; Ichiro Fujisawa; Toshiaki Sano (December 2009). "Pituitary and stalk lesions (infundibulo-hypophysitis) associated with immunoglobulin G4-related systemic disease: an emerging clinical entity". Endocrine Journal. 56 (9): 1033–1041. doi:10.1507/endocrj.K09E-277. PMID 19926920.
- ↑ Takahira, Masayuki; Azumi, Atsushi (21 November 2013). "Chapter 12: Ophthalmology". In Hisanori Umehara; Kazuichi Okazaki; John H. Stone; Shigeyuki Kawa; Mitsuhiro Kawano (eds.). IgG4-Related Disease. Springer Science & Business Media. pp. 77–84. doi:10.1007/978-4-431-54228-5. ISBN 978-4-431-54227-8. Archived from the original on 26 August 2017. Retrieved 7 May 2021.
- ↑ Jiwon Seo; In Ji Song; Sak Lee; Hyeon Joo Jeong; Hye Min Kim; Beom Seok Koh; Sung Ha Park (March 2015). "A Case of Constrictive Pericarditis due to Immunoglobulin G4-Related Disease". Korean Circulation Journal. 45 (2): 161–164. doi:10.4070/kcj.2015.45.2.161. PMC 4372983. PMID 25810739.
- ↑ 13.0 13.1 13.2 Changho Song; Myoung Ju Koh; Yong-Nam Yoon; Boyoung Joung; Se Hoon Kim (1 September 2013). "IgG4-related sclerosing disease involving the superior vena cava and the atrial septum of the heart". Yonsei Medical Journal. 54 (5): 1285–1288. doi:10.3349/ymj.2013.54.5.1285. PMC 3743176. PMID 23918583.
- ↑ Qi Qian; Kianoush B. Kashani; Dylan V. Miller (10 September 2009). "Ruptured abdominal aortic aneurysm related to IgG4 periaortitis". The New England Journal of Medicine. 361 (11): 1121–1123. doi:10.1056/NEJMc0905265. PMID 19741239.
- ↑ Satomi Kasashima; Yoh Zen (26 January 2011). "IgG4-related Inflammatory Abdominal Aortic Aneurysm, Spectrum of IgG4-related Chronic Periaortitis". Annals of Vascular Diseases. 3 (3): 182–189. doi:10.3400/avd.sasvp01002. PMC 3595781. PMID 23555408.
- ↑ John H. Stone; Arezou Khosroshahi; Alan Hilgenberg; Amy Spooner; Eric M. Isselbacher; James R. Stone (October 2009). "IgG4-related systemic disease and lymphoplasmacytic aortitis". Arthritis & Rheumatism. 60 (10): 3139–3145. doi:10.1002/art.24798. PMID 19790067.
- ↑ Andrea Barp; Marny Fedrigo; Filippo Maria Farina; Sandro Lepidi; Francesco Causin; Chiara Castellani; Giacomo Cester; Thiene; Marialuisa Valente; Claudio Baracchini; Annalisa Angelini (January–February 2016). "Carotid aneurism with acute dissection: an unusual case of IgG4-related diseases". Cardiovascular Pathology. 25 (1): 59–62. doi:10.1016/j.carpath.2015.08.006. PMID 26453089. Archived from the original on 2021-08-28. Retrieved 2021-05-07.
- ↑ Inoue, Dai; Yoshida, Kotaro; Yoneda, Norihide; Ozaki, Kumi; Matsubara, Takashi; Nagai, Keiichi; Okumura, Kenichirou; Toshima, Fumihito; Toyama, Jun (April 2015). "IgG4-Related Disease: Dataset of 235 Consecutive Patients". Medicine. 94 (15): e680. doi:10.1097/MD.0000000000000680. PMC 4602507. PMID 25881845.
- ↑ Guillermo Delgado-García; Sergio Sánchez-Salazar; Erick Rendón-Ramírez; Mario Castro-Medina; Bárbara Sáenz-Ibarra; Álvaro Barboza-Quintana; María Azalea Loredo-Alanis; David Hernández-Barajas; Dionicio Galarza-Delgado (November 2016). "Myocardial ischemia as presenting manifestation of IgG4-related disease: a case-based review". Clinical Rheumatology. 35 (11): 2857–2864. doi:10.1007/s10067-016-3292-z. PMID 27142374. S2CID 19891657.
- ↑ Paulo Sampaio Gutierrez; Thiago Schultz; Sheila Aparecida Coelho Siqueira; Luciano de Figueiredo Borges (November–December 2013). "Sudden coronary death due to IgG4-related disease". Cardiovascular Pathology. 22 (6): 505–507. doi:10.1016/j.carpath.2013.05.003. PMID 23830124. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ Brittany J. Holmes; Nikolay G. Delev; Gary R. Pasternack; Marc K. Halushka (12 June 2012). "Novel cause of sudden cardiac death: IgG4-related disease". Circulation. 125 (23): 2956–2957. doi:10.1161/CIRCULATIONAHA.111.061002. PMID 22689933.
- ↑ Edward M. Kobraei; Tae H. Song; Douglas J. Mathisen; Vikram Deshpande; Eugene J. Mark (October 2013). "Immunoglobulin G4-Related Disease Presenting as an Obstructing Tracheal Mass: Consideration of Surgical Indications". The Annals of Thoracic Surgery. 96 (4): e91–e93. doi:10.1016/j.athoracsur.2013.04.120. PMID 24088500. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ 23.0 23.1 Hirotsugu Ohkubo; Mikinori Miyazaki; Tetsuya Oguri; Atsushi Arakawa; Yoichiro Kobashi; Akio Niimi (June 2015). "A rare case of IgG4-related disease involving the uterus". Rheumatology. 54 (6): 1124–1125. doi:10.1093/rheumatology/kev024. PMID 25810424.
- ↑ Jong Hyun Choi; Jae Kyeom Sim; Jee Youn Oh; Eun Joo Lee; Gyu Young Hur; Seung Heon Lee; Sung Yong Lee; Je Hyeong Kim; Sang Yeub Lee; Chol Shin; Jae Jeong Shim; Kwang Ho In; Kyung Ho Kang; Kyung Hoon Min (April 2014). "A Case of IgG4-Related Disease Presenting as Massive Pleural Effusion and Thrombophlebitis". Tuberculosis and Respiratory Diseases. 76 (4): 179–183. doi:10.4046/trd.2014.76.4.179. PMC 4021266. PMID 24851132.
- ↑ 25.0 25.1 Hyuk Lee; Mee Joo; Tae Jun Song; Sun Hee Chang; Hanseong Kim; Yeon Soo Kim; Ji Yoon Ryoo (April 2011). "IgG4-related sclerosing esophagitis: a case report". Gastrointestinal Endoscopy. 73 (4): 834–837. doi:10.1016/j.gie.2010.08.043. PMID 21067736. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ 26.0 26.1 Myriam Dumas-Campagna; Simon Bouchard; Genevieve Soucy; Mickael Bouina (August 2014). "IgG4-related esophageal disease presenting as esophagitis dissecans superficialis with chronic strictures". Journal of Clinical Medicine Research. 6 (4): 295–298. doi:10.14740/jocmr1845w. PMC 4039102. PMID 24883156.
- ↑ Bruno Coulier; Luc Montfort; Gabriela Beniuga; Frederic Pierard; Isabelle Gielen (January–February 2014). "Small Bowel Obstruction Caused by Peritoneal Immunoglobulin G4-Related Disease Mimicking Carcinomatosis: Case Report". Korean Journal of Radiology. 15 (1): 66–71. doi:10.3348/kjr.2014.15.1.66. PMC 3909864. PMID 24497794.
- ↑ 28.0 28.1 Yassaman Raissian; Samih H. Nasr; Christopher P. Larsen; Robert B. Colvin; Thomas C. Smyrk; Naoki Takahashi; Ami Bhalodia; Aliyah R. Sohani; Lizhi Zhang; Suresh Chari; Sanjeev Sethi; Mary E. Fidler; Lynn D. Cornell (July 2011). "Diagnosis of IgG4-Related Tubulointerstitial Nephritis". Journal of the American Society of Nephrology. 22 (7): 1343–1352. doi:10.1681/ASN.2011010062. PMC 3137582. PMID 21719792. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ Satoshi Yazawa; Rei Ohara; Takahiro Maeda; Kent Kanao; Seiya Hattori; Yosuke Nakajima; Mototsugu Oya (July 2013). "[Three cases of IgG4-related disease associated with urinary tract obstruction]". Nihon Hinyokika Gakkai Zasshi (The Japanese Journal of Urology) (in 日本語). 104 (4): 620–625. doi:10.5980/jpnjurol.104.620. PMID 23971373.
- ↑ 30.0 30.1 Bobby B Najari; Brian D Robinson; Stephen A Paget; Darius A Paduch (October 2014). "Clinical, radiographic, and pathologic description of IgG4-related perivasal fibrosis: a previously undescribed etiology of chronic orchialgia". Urology. 84 (4): 748–750. doi:10.1016/j.urology.2014.06.035. PMID 25260440. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ 31.0 31.1 31.2 Arezou Khosroshahi; John H. Stone (January 2011). "A clinical overview of IgG4-related systemic disease". Current Opinion in Rheumatology. 23 (1): 57–66. doi:10.1097/BOR.0b013e3283418057. PMID 21124086. S2CID 45093804.
- ↑ 32.0 32.1 John H. Stone; Yoh Zen; Vikram Deshpande (February 2012). "IgG4-Related Disease". The New England Journal of Medicine. 366 (6): 539–51. doi:10.1056/NEJMra1104650. PMID 22316447.
- ↑ 33.0 33.1 33.2 33.3 Herwig Pieringer; Ilse Parzer; Adelheid Wöhrer; Petra Reis; Bastian Oppl; Jochen Zwerina (16 July 2014). "IgG4- related disease: an orphan disease with many faces". Orphanet Journal of Rare Diseases. 9: 110. doi:10.1186/s13023-014-0110-z. PMC 4223520. PMID 25026959.
- ↑ 34.0 34.1 Tetsuo Himi; Kenichi Takano; Motohisa Yamamoto; Yasuyoshi Naishiro; Hiroki Takahashi (February 2012). "A novel concept of Mikulicz's disease as IgG4-related disease". Auris Nasus Larynx. 39 (1): 9–17. doi:10.1016/j.anl.2011.01.023. PMID 21571468. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ 35.0 35.1 Yueyang Guo; David Ansdell; Sharon Brouha; Andrew Yen (January 2015). "Coronary periarteritis in a patient with multi-organ IgG4-related disease". Journal of Radiology Case Reports. 9 (1): 1–17. doi:10.3941/jrcr.v9i1.1967. PMC 4391803. PMID 25926916.
- ↑ Vikram Deshpande; Arezou Khosroshahi; Gunnlaugur P Nielsen; Daniel L Hamilos; John H Stone (May 2011). "Eosinophilic angiocentric fibrosis is a form of IgG4-related systemic disease". The American Journal of Surgical Pathology. 35 (5): 701–706. doi:10.1097/PAS.0b013e318213889e. PMID 21502911. S2CID 21074120.
- ↑ Mollie N. Carruthers; Eli M. Miloslavsk; John H. Stone (August 2013). "Reply to "IgG4-related pharyngitis-an addition to the nomenclature of IgG4-related disease: comment on the article by Stone et al."". Arthritis & Rheumatism. 65 (8): 2217–2218. doi:10.1002/art.37998. PMID 23677887.
- ↑ Wah Cheuk; Fiona K Y Tam; Alice N H Chan; Ivy S C Luk; Anthony P W Yuen; Wai-Kong Chan; Terry C W Hung; John K C Chan (November 2010). "Idiopathic cervical fibrosis--a new member of IgG4-related sclerosing diseases: report of 4 cases, 1 complicated by composite lymphoma". The American Journal of Surgical Pathology. 34 (11): 1678–1685. doi:10.1097/PAS.0b013e3181f12c85. PMID 20871392. S2CID 364195.
- ↑ Shyamal H. Mehta; Jeffrey A. Switzer; Paul Biddinger; Amyn M. Rojiani (11 February 2014). "IgG4-related leptomeningitis: a reversible cause of rapidly progressive cognitive decline". Neurology. 82 (6): 540–542. doi:10.1212/WNL.0000000000000100. PMID 24384648. S2CID 207106461.
- ↑ Akiyuki Hiraga; Daisuke Ozaki; Atsuko Tsuneyama; Seiro Ito; Kyosuke Koide; Satoshi Kuwabara (15 October 2015). "Corticosteroid-responsive leptomeningitis with IgG4-positive plasma-cell infiltration". Journal of the Neurological Sciences. 357 (1–2): 338–340. doi:10.1016/j.jns.2015.07.048. PMID 26254623. S2CID 1062402. Archived from the original on 21 September 2021. Retrieved 7 May 2021.
- ↑ Terumi Kamisawa; Yuyang Tu; Naoto Egawa; Nobuhiro Sakaki; Jun-ichi Ishiwata; Kouji Tsuruta; Atsutake Okamoto; Toshio Takahashi; Morio Koike (January 2001). "[Clinicopathologic study on chronic pancreatitis with diffuse irregular narrowing of the main pancreatic duct]". Nippon Shokakibyo Gakkai Zasshi (The Japanese Journal of Gastroenterology) (in 日本語). 98 (1): 15–24. doi:10.11405/nisshoshi1964.98.15. PMID 11201120.
- ↑ 42.0 42.1 Dai Inoue; Yoh Zen; Hitoshi Abo; Toshifumi Gabata; Hiroshi Demachi; Jyun Yoshikawa; Shiro Miyayama; Yasuni Nakanuma; Osamu Matsui (November 2011). "Immunoglobulin G4–related Periaortitis and Periarteritis: CT Findings in 17 Patients". Radiology. 261 (2): 625–633. doi:10.1148/radiol.11102250. PMID 21803920.
- ↑ 43.0 43.1 43.2 Verghese George; Varaha Sairam Tammisetti; Venkateswar Rao Surabhi; Alampady K. Shanbhogue (July 2013). "Chronic Fibrosing Conditions in Abdominal Imaging". RadioGraphics. 33 (4): 1053–1080. doi:10.1148/rg.334125081. PMID 23842972.
- ↑ Jin Hee Kim; Jae Ho Byun; Seung Soo Lee; Hyoung Jung Kim; Moon-Gyu Lee (January 2013). "Atypical Manifestations of IgG4-Related Sclerosing Disease in the Abdomen: Imaging Findings and Pathologic Correlations". American Journal of Roentgenology. 200 (1): 102–111. doi:10.2214/AJR.12.8783. PMID 23255748.
- ↑ Takashi Karashima; Yoshinori Taniguchi; Tsutomu Shimamoto; Tomoya Nao; Hiroshi Nishikawa; Satoshi Fukata; Masayuki Kamada; Keiji Inoue; Kentaro Oko; Hideki Nakajima; Shigetoshi Sano; Manabu Matsumoto; Naoto Kuroda; Yoshihiro Kamei; Taro Shuin (9 December 2014). "IgG4-related disease of the paratestis in a patient with Wells syndrome: a case report". Diagnostic Pathology. 9: 225. doi:10.1186/s13000-014-0225-5. PMC 4265405. PMID 25487870.
- ↑ Hans Bösmüller; Claus Hann von Weyhern; Patrick Adam; Vedat Alibegovic; Gregor Mikuz; Falko Fend (January 2011). "Paratesticular fibrous pseudotumor--an IgG4-related disorder?". Virchows Archiv. 458 (1): 109–113. doi:10.1007/s00428-010-0995-4. PMID 20957491. S2CID 21848271.
- ↑ Yasuhito Hamaguchi; Manabu Fujimoto; Yukiyo Matsushita; Seiko Kitamura-Sawada; Mitsuhiro Kawano; Kazuhiko Takehara (2011). "IgG4-related skin disease, a mimic of angiolymphoid hyperplasia with eosinophilia". Dermatology. 223 (4): 301–305. doi:10.1159/000335372. PMID 22269779. S2CID 22928854.
- ↑ Yoshiki Tokura; Hiroaki Yagi; H. Yanaguchi; Yuta Majima; Akira Kasuya; Taisuke Ito; M Maekawa; Hideo Hashizume (November 2014). "IgG4-related skin disease". British Journal of Dermatology. 171 (5): 959–967. doi:10.1111/bjd.13296. PMID 25065694. S2CID 5374017.
- ↑ Dai Inoue; Yoh Zen; Yasuharu Sato; Hitoshi Abo; Hiroshi Demachi; Akio Uchiyama; Toshifumi Gabata; Osamu Matsui (2012). "IgG4-Related Perineural Disease". International Journal of Rheumatology. 2012: 401890. doi:10.1155/2012/401890. PMC 3317227. PMID 22523496.
- ↑ Nicholas Andrew; Daniel Kearney; Nicole Sladden; Alastair Goss; Dinesh Selva (April 2014). "Immunoglobulin G4-related disease of the hard palate". Journal of Oral and Maxillofacial Surgery. 72 (4): 717–723. doi:10.1016/j.joms.2013.08.033. PMID 24268962. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ Katie E. Rollins; Samir P. Mehta; Maria O'Donovan; Peter M. Safranek (2011). "Gastric IgG4-Related Autoimmune Fibrosclerosing Pseudotumour: A Novel Location". ISRN Gastroenterology. 2011 (2011): 873087. doi:10.5402/2011/873087. PMC 3168566. PMID 21991533.
- ↑ Younghwan Ko; Ji Young Woo; Jeong Won Kim; Hye Sook Hong; Ik Yang; Yul Lee; Daehyun Hwang; Seon Jeong Min (Sep–Oct 2013). "An Immunoglobulin G4-Related Sclerosing Disease of the Small Bowel: CT and Small Bowel Series Findings". Korean Journal of Radiology. 14 (5): 776–780. doi:10.3348/kjr.2013.14.5.776. PMC 3772257. PMID 24043971.
- ↑ Sung-Bong Choi; Chul-Hyun Lim; Myung-Guen Cha; Won-Kyung Kang (2 May 2016). "IgG4-related disease of the rectum". Annals of Surgical Treatment and Research. 90 (5): 292–295. doi:10.4174/astr.2016.90.5.292. PMC 4865708. PMID 27186575.
- ↑ C. Costa; C. Saraiva; S. Freitas (25 September 2013). "Adrenal lesion as first manifestation of IgG4-related sclerosing disease". EuroRad. European Society of Radiology. doi:10.1594/EURORAD/CASE.11230. Retrieved 15 May 2015.[permanent dead link]
- ↑ Miroslav Sekulic; Simona Pichler Sekulic; Saeid Movahedi-Lankarani (4 May 2016). "IgG4-related Disease of the Ovary: A First Description". International Journal of Gynecological Pathology. 36 (2): 190–194. doi:10.1097/PGP.0000000000000293. PMID 27149005.
- ↑ Sun A. Kim; Sang-Ryung Lee; Jooryung Huh; Steven S. Shen; Jae Y. Ro (August 2011). "IgG4-associated inflammatory pseudotumor of ureter: clinicopathologic and immunohistochemical study of 3 cases". Human Pathology. 42 (8): 1178–1184. doi:10.1016/j.humpath.2010.03.011. PMID 21334715.
- ↑ Sanghui Park; Jae Y. Ro; Dong Hyeon Lee; Sun Young Choi; Heasoo Koo (December 2013). "Immunoglobulin G4-associated inflammatory pseudotumor of urinary bladder: a case report". Annals of Diagnostic Pathology. 17 (6): 540–543. doi:10.1016/j.anndiagpath.2013.01.004. PMID 23434261. Archived from the original on 2021-09-21. Retrieved 2021-05-07.
- ↑ Travis W. Dum; Da Zhang; Eugene K. Lee (2015). "IgG4-Related Disease in a Urachal Tumor". Case Reports in Urology. 2014 (2015): 275850. doi:10.1155/2014/275850. PMC 4151357. PMID 25202466.
- ↑ Koichiro Shinoda; Shoko Matsui; Hirofumi Taki; Hiroyuki Hounoki; Reina Ogawa; Shin Ishizawa; Kazuyuki Tobe (January 2011). "Deforming arthropathy in a patient with IgG4-related systemic disease: Comment on the article by Stone et al". Arthritis Care & Research. 63 (1): 172. doi:10.1002/acr.20297. PMID 20662046. S2CID 205221152.
- ↑ Takao Taniguchi; Hisato Kobayashi; Shouichi Fukui; Keiji Ogura; Tatuyoshi Saiga; Motozumi Okamoto (September 2006). "A case of multifocal fibrosclerosis involving posterior mediastinal fibrosis, retroperitoneal fibrosis, and a left seminal vesicle with elevated serum IgG4". Human Pathology. 37 (9): 1237–1239. doi:10.1016/j.humpath.2006.03.021. PMID 16938531.
- ↑ Gerhard Seifert; Karl Donath (March 1977). "Zur Pathogenese des Küttner-Tumors der Submandibularis - Analyse von 347 Fällen mit chronischer Sialadenitis der Submandibularis" [On the pathogenesis of the Küttner tumor of the submandibular gland - Analysis of 349 cases with chronic sialadenitis of the submandibular (author's translation)]. HNO (in Deutsch). 25 (3): 81–92. PMID 856776.
- ↑ Zachary S. Wallace; Hamid Mattoo; Vinay S. Mahajan; Maria Kulikova; Leo Lu1; Vikram Deshpande; Hyon K. Choi; Shiv Pillai; John H. Stone (6 June 2016). "Predictors of disease relapse in IgG4-related disease following rituximab". Rheumatology. 55 (6): 1000–1008. doi:10.1093/rheumatology/kev438. PMC 4900135. PMID 26888853.
- ↑ Cory A. Perugino; John H. Stone (September 2016). "Treatment of IgG4-related disease: Current and future approaches". Zeitschrift für Rheumatologie. 75 (7): 681–686. doi:10.1007/s00393-016-0142-y. PMC 5953192. PMID 27431746.
- ↑ "Xencor Initiates Two Phase 2 Trials of XmAb5871 in IgG4-Related Disease and Systemic Lupus Erythematosus". PR Newswire. 7 March 2016. Archived from the original on 19 August 2016. Retrieved 23 July 2016.
The primary objective of the study is to evaluate the effect of every other week IV administration of XmAb5871 on the IgG4-RD Responder Index (RI) in patients with active IgG4-RD.
- ↑ "Final Results of an Open Label Phase 2 Study of a Reversible B Cell Inhibitor, Xmab®5871, in IgG4-Related Disease - ACR Meeting Abstracts". ACR Meeting Abstracts. Archived from the original on 2020-10-30. Retrieved 2021-05-07.
- ↑ 66.0 66.1 66.2 Hisanori Umehara; Kazuichi Okazaki; Yasufumi Masaki; Mitsuhiro Kawano; Motohisa Yamamoto; Takako Saeki; Shoko Matsui; Takayuki Sumida; Tsuneyo Mimori; Yoshiya Tanaka; Kazuo Tsubota; Tadashi Yoshino; Shigeyuki Kawa; Ritsuro Suzuki; Tsutomu Takegami; Naohisa Tomosugi; Nozomu Kurose; Yasuhito Ishigaki; Atsushi Azumi; Masaru Kojima; Shigeo Nakamura; Dai Inoue; The Research Program for Intractable Disease by Ministry of Health, Labor and Welfare Japan G4 team (February 2012). "A novel clinical entity, IgG4-related disease (IgG4RD): general concept and details". Modern Rheumatology. 22 (1): 1–14. doi:10.1007/s10165-011-0508-6. PMC 3278618. PMID 21881964.
- ↑ "Clinical Commissioning Policy: Rituximab for immunoglobulin G4-related disease (IgG4-RD)" (PDF). NHS England. December 2016. Archived (PDF) from the original on 2 February 2017. Retrieved 27 January 2017.
External links[edit | edit source]
| Classification | |
|---|---|
| External resources |
- Overview of IgG4-related disease Archived 2020-09-23 at the Wayback Machine - UpToDate's article on IgG4-related disease.
- DermNet NZ Archived 2016-06-04 at the Wayback Machine entry
Categories: [IgG4-related disease] [Immune system disorders] [Autoimmune diseases] [Pancreas disorders] [Salivary gland pathology] [Hepatology] [Steroid-responsive inflammatory conditions]
↧ Download as ZWI file | Last modified: 10/27/2023 16:37:09 | 1 views
☰ Source: https://mdwiki.org/wiki/IgG4-related_disease | License: CC BY-SA 3.0
 ZWI signed:
ZWI signed:.jpg)
.jpg)
 KSF
KSF