Postpartum Psychosis
 From Mdwiki
From Mdwiki | Postpartum psychosis | |
|---|---|
| Other names: Puerperal psychosis, peripartum psychosis | |
 | |
| Rate and timing of onset of psychoses among Swedish first-time mothers | |
| Specialty | Obstetrics, psychiatry |
| Symptoms | Hallucinations, delusions, mood swings, confusion, restlessness, personality changes[1] |
| Complications | Suicide, infanticide[2] |
| Usual onset | Within 2 weeks of delivery[3] |
| Duration | 1 to 12 months[2] |
| Causes | Unclear[1] |
| Risk factors | First pregnancies, older age, loss of sleep, personal or family history of the condition or bipolar disorder[4][3] |
| Diagnostic method | Based on symptoms after ruling out other potential causes[2] |
| Differential diagnosis | Normal adjustment after birth, postpartum depression, post partum bleeding, endometritis, drug intoxication, drug withdrawal, autoimmune thyroiditis, autoimmune encephalitis, postpartum obsessive-compulsive disorder[3][4] |
| Treatment | Anti-psychotics, lithium, benzodiazepines, electroconvulsive therapy (ECT)[3][4] |
| Frequency | 1 to 2 per 1000 childbirths[3][4] |
Postpartum psychosis (PPP) involves the abrupt onset of psychotic symptoms shortly following childbirth, typically within two weeks of delivery but less than 4 weeks.[3][4] Symptoms may include seeing or smelling things that are not there (hallucinations), thoughts that are unlikely to be true (delusions), abnormally elevated mood (mania), depression, anxiety, or confusion.[1] Other symptoms may include disorganized thoughts, difficulty sleeping, and variable mood and consciousness.[3][4] There is generally little understanding of the severity of the condition by the person themselves.[4] Over half of those affected are later diagnosed with bipolar.[4]
The cause is unknown, though may relate to hormone changes, immune system problems, and genetics.[1][4][3] Risk factors include first pregnancies, older age, loss of sleep, and a personal or family history of PPP or bipolar disorder.[4][3] Life stress does not appear to play a role, nor is evidence clear around complications of pregnancy.[2] There are no screening or assessment tools for diagnosis;[3] which is made based on the presenting symptoms, guided by criteria in the DSM-V after ruling out other potential causes.[2] In the Diagnostic and Statistical Manual of Mental Disorders, Volume V (DSM-V TR) it is under brief psychotic disorder with the specifier "peripartum onset", rather then as a separate entity.[5][2]
It is considered a psychiatric emergency requiring urgent hospitalization.[3] Treatment may include medications such as lithium, benzodiazepines, and antipsychotics, as well as the procedure electroconvulsive therapy (ECT).[3][4] In cases where a women has previously been effected lithium may be started preventatively immediately after delivery.[4] Psychotic symptoms, particularly those that include delusions of misidentification, cause safety concerns for the baby and mother;[6] with the condition resulting in the child's death in 4% of cases and a risk of suicide by the mother.[4] Divorce occurs in up to 20%.[2]
Postpartum psychosis occurs in 1 to 2 per 1000 childbirths.[3][4] Rates appear similar in different cultures and social classes.[2] More frequently, it occurs in the context of known or new-onset bipolar, known as postpartum bipolar disorder.[2] The condition has been described since 400 BC by Hippocrates.[3]
Signs and symptoms[edit | edit source]

By its diagnostic definition (under the name "brief psychotic disorder with peripartum onset"), PPP occurs either during pregnancy or within 4 weeks of delivery.[7] Generally, symptoms have been observed within 3–10 days of childbirth, though women with a past history of bipolar disorder may experience symptoms even sooner.[3][2] The persistence of symptoms varies; while the average reported length of an episode may last weeks to several months,[2] there is currently no strong literature documenting the course of individual episodes. Recurrence rates for psychotic episodes, on the other hand, have stronger supporting studies.
Diagnostic criteria per the DSM-V require the presence of at least one psychotic symptom, defined as delusions, hallucinations, bizarre or incoherent speech (disorganized speech), or abnormal movements (psychomotor behavior) such as catatonia.[7] Delusions, particularly about the infant, are the most commonly reported psychotic symptom associated with PPP.[4][2][8] Paranoid delusions are a frequently noted theme in cases of PPP,[4][9] but a small review noted infrequent cases of delusional misidentification syndromes, such as Capgras syndrome (the belief that someone or something familiar has been replaced with an impostor), Fregoli syndrome (the belief that a stranger is actually a known person in disguise), and others.[6] The latter types of delusions may have a negative impact on the bond between a mother and child, raising concerns for the safety of both.[6][10]
Both postpartum obsessive-compulsive disorder (OCD) and PPP may present with concerning thoughts about the infant; typically, the thoughts associated with OCD are unwanted and distressing to the individual (who does not wish to act on their thoughts), whereas persons with PPP are often less distressed by their beliefs and may even feel the need to act on them.[3][4][11] Compared to schizophrenia, PPP tends to feature less bizarre delusions, and associated hallucinations are more likely to be visual rather than auditory.[2] The sensation of being outside one's body or feeling that one's surroundings are unreal (i.e., derealization) has also been described in cases of PPP.[3] Additional distinctions in PPP compared to classical schizophrenia include the presence of mood and cognitive (or neurological) symptoms.[3][2]
Rapid mood changes or the presence of abnormal moods such as depression or mania (increased energy, decreased need for sleep, etc.) tend to be seen in a large percentage of patients experiencing PPP.[3][4][2] Irritability, anxiety, and general difficulties with sleep may also be present.[2] Confusion or disorientation, disorganized thoughts, incoherent speech, or abrupt changes in a person's mental capacity may also be seen in individuals experiencing PPP,[4][2] though one small study observed these neurological symptoms in only one-quarter of PPP cases.[2][12] Like delirium, these symptoms may come and go in unpredictable patterns.[2]
Thoughts of committing suicide or harming one's infant or children have also been reported as common occurrences in PPP, with as many as half of PPP cases exhibiting these features.[3][2] In many cases where harmful thoughts exist, the person experiencing these thoughts does not consider their intended action to be harmful; rather, they believe that their actions are in the best interest of the child.[4][2]
In addition to the rapid onset of symptoms (less than two weeks) with the presence of a psychotic symptom, further diagnostic criteria defined by the DSM-V for "brief psychotic disorder with peripartum onset" include that the symptomatic episode ends within one month and involves a return to the individual's previous functional ability, as well as confidence that the episode is not a different psychiatric illness (e.g., depressive or bipolar disorder with psychotic features) or the result of substance-induced psychosis.[7]
Risk factors[edit | edit source]
Childbirth is the primary cause of PPP; other causes and risk factors remain largely under investigation.[2]
The largest known risks for the occurrence of PPP include a history of PPP in a previous pregnancy, or a personal or family history of bipolar disorder.[3][2] A significant number of PPP cases, however, occur in individuals with no prior history of psychosis.[2][13] (For this reason, first-time pregnancy is itself sometimes considered to be a risk factor for PPP.)[3][4][2] A review of pregnancy-related complications demonstrated some association between emergency caesarean sections (C-sections), excess bleeding, uterine rupture, and stillbirth (amongst other complications) and the subsequent development of PPP; however, several of the reviewed studies were contradictory and thus no consensus can confirm the relationship between problems related to pregnancy and PPP.[2][14]
Lifestyle and psychological factors, such as previous trauma or single parenthood, have likewise been inconclusive as factors contributing to PPP,[8] though a number of patients have reported a perception that social and pregnancy-related challenges were the cause of their PPP episodes.[8][15]
Pathophysiology[edit | edit source]
Currently, the pathophysiology of PPP is not well understood and remains an open field of ongoing research. The leading theories under investigation involve areas of genetics, hormones, immunology, and sleep disturbance processes.[2]
Genetics[edit | edit source]
Some findings suggest an association between PPP and variation in serotonin transporter genes and signaling or changes at specific chromosomes (e.g., 16p13 or METTL13). However, the majority of research devoted to genetic understanding of PPP has evaluated patients who have known bipolar disorder, so these associations may not be specific to PPP.[2][9][16][17][18][19]
Hormones[edit | edit source]
Despite significant hormone changes that occur around pregnancy and childbirth, there is little evidence supporting hormonal causes behind PPP.[2][9] Changes in corticotropin-releasing hormone (CRH) and adrenocorticotropic hormone (ACTH), as well as rapid changes in estrogen and progesterone, are known effects associated with delivering a child, and they are present in both those affected and unaffected by PPP; therefore, a relationship between hormonal change and the onset of psychosis is not well-supported,[2][9] though some researchers continue to explore whether postpartum disorders might be related to differences in sensitivity to rapid hormone changes.[20] Estrogen has known impacts on various neurotransmitters, including serotonin and dopamine),[21] which prominent theories associate with schizophrenia;[22] however, investigations of estrogen concentrations and dopamine receptor sensitivity, and trials of estrogen replacement following birth, do not support an association between estrogen changes and PPP onset.[9]
Immune[edit | edit source]
Due to above-average rates of PPP in individuals experiencing immunologic complications such as anti-N-methyl-D-aspartate (NMDA) receptor encephalitis and autoimmune thyroid disorders,[23][2] as well as the known heightened immune response post-childbirth, some theories suggest a connection between PPP and the immune system.[2][24] There is some evidence connecting PPP with changes to levels of peripheral immune cells (e.g., lymphocytes and NK or natural killer cells) traveling in the bloodstream, but more research is required to identify the specific mechanisms and cell types involved which might be related to PPP onset.[23] No direct evidence has shown a link between cytokine levels and PPP.[23]
Sleep disturbance[edit | edit source]
A link between sleep difficulty and PPP is not strongly supported by current evidence; however, some studies have demonstrated an increased risk for postpartum psychosis in women with bipolar disorder who have had manic episodes triggered by sleep disturbance.[2][25]
Diagnosis[edit | edit source]
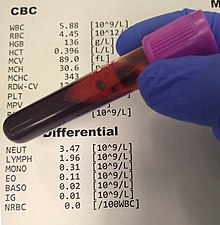
Not recognized as its own distinct disorder, PPP is instead classified by the DSM-V as a "Brief Psychotic Disorder with peripartum onset."[7][2] Clinical requirements for the diagnosis of a brief psychotic disorder require the presence of at least one of the following psychotic symptoms: delusions, hallucinations, disorganized speech, and/or grossly disorganized or catatonic behavior.[7] Additional requirements include that the psychotic symptom lasts between one day and one month, eventually resulting in the person recovering their previous level of functional ability, and that the symptoms are not better related to a different psychiatric illness (including the result of ingesting substances such as alcohol or drugs).[7] The specifier, "with peripartum onset," requires the development of the above within 4 weeks of delivering a child.[7]
There are no laboratory or imaging tools available to diagnose PPP, though a work-up of different laboratory analyses and imaging of the brain may be conducted to ensure other potential confounding diagnoses (e.g., vascular disorders, infective delirium, etc.) are not the cause of the patient's presentation.[2] These may include, but are not limited to, a complete blood count, comprehensive metabolic panel, urinalysis and urine drug screen, and tests for thyroid functioning; further workup in the setting of classically neurological symptoms (such as delirium-like confusion) may include magnetic resonance imaging (MRI), a test of cerebrospinal fluid (CSF), or electroencephalogram (EEG).[2]
Screening[edit | edit source]
Currently, no screening tools exist to evaluate for PPP, though providers may choose to use standard screens for postpartum depression and mania to evaluate the presence of these particular symptoms.[2]
It may be difficult for providers and family-members to identify PPP, due to the presence of several symptoms that are classically associated with other postpartum conditions such as postpartum depression, as well as overlap with often-benign changes that accompany new parenthood (anxiety, irritability, poor sleep). [4][2] Therefore, clinical recommendations advise healthcare providers to directly ask new mothers about thoughts of harming themselves or their children.[3]
Differential[edit | edit source]
The presentation of PPP includes a large differential overlapping neurological and psychiatric diseases and syndromes. Neurological, or cognitive, symptoms like disorientation and confusion raise concerns for conditions that organically affect the brain: this may include autoimmune processes resulting in various forms of encephalitis, embolism (and other vascular disorders), and infectious processes.[7][2] Physical causes as a result of chronic hormonal disease (e.g., thyroid diseases, hyperparathyroidism) or complications resulting from childbirth, such as Sheehan's syndrome or other complications resulting from excess blood loss, should also be evaluated.[4][2] Other psychiatric conditions must also be considered: postpartum blues, postpartum depression, anxiety disorders, and postpartum OCD may have many overlapping symptoms with PPP.[2] Finally, psychosis as a result of various substances (including medications such as steroids), should be ruled out.[2] If this is a first-onset episode of psychosis, new-onset bipolar disorder and schizophrenia cannot be ruled out; the diagnosis of these disorders is based on time and recurrence of episodes.[7]
Other non-organic[edit | edit source]
It is much less common to encounter other acute psychoses are the time of delivery.
Psychogenic psychosis[edit | edit source]
This is the name given to a psychosis whose theme, onset and course are all related to an extremely stressful event.[26] The psychotic symptom is usually a delusion. Over 50 cases have been described, but usually in unusual circumstances, such as abortion[27] or adoption,[28] or in fathers at the time of the birth of one of their children.[29] They are occasionally seen after normal childbirth.[30]: 169–171
Paranoid and schizophrenic psychoses[edit | edit source]
These are so uncommon in the puerperium that it seems reasonable to regard them as sporadic events, not puerperal complications.
Early postpartum stupor[edit | edit source]
Brief states of stupor have rarely been described in the first few hours or days after the birth.[30]: 119–122 They are similar to parturient delirium and stupor, which are among the psychiatric disorders of childbirth.
Organic psychoses[edit | edit source]
There are at least a dozen organic (neuropsychiatric) psychoses that can present in pregnancy or soon after childbirth.[30]: 200–204 The clinical picture is usually delirium – a global disturbance of cognition, affecting consciousness, attention, comprehension, perception and memory – but amnesic syndromes and a mania-like state[30]: 25 also occur. The two most recent were described in 1980[31] and 2010,[32] and it is quite likely that others will be described. Organic psychoses, especially those due to infection, may be more common in nations with high parturient morbidity.[33]
Infective delirium[edit | edit source]
The most common organic postpartum psychosis is infective delirium. This was mentioned by Hippocrates:[34] there are 8 cases of puerperal or post-abortion sepsis among the 17 women in the 1st and 3rd books of epidemics, all complicated by delirium. In Europe and North America the foundation of the metropolitan maternity hospitals, together with instrumental deliveries and the practice of attending necropsies, led to epidemics of streptococcal puerperal fever, resulting in maternal mortality rates up to 10%. The peak was about 1870, after which antisepsis and asepsis gradually brought them under control. These severe infections were often complicated by delirium, but it was not until the nosological advances of Chaslin[35] and Bonhöffer[36] that they could be distinguished from other causes of postpartum psychosis. Infective delirium hardly ever starts during pregnancy, and usually begins in the first postpartum week. The onset of sepsis and delirium are closely related, and the course parallels the infection, although about 20% of patients continue to have chronic confusional states after recovery from the infection. Recurrences after another pregnancy are rare. Their frequency began to decline at the end of the 19th century,[37] and fell steeply after the discovery of the sulphonamides. Puerperal sepsis is still common in Bangladesh,[38] Nigeria[39] and Zambia.[40] Even in the United Kingdom, cases are still occasionally seen.[30]: 27 It would be a mistake to forget this cause of puerperal psychosis.
Eclamptic and Donkin psychoses[edit | edit source]
Eclampsia is the sudden eruption of convulsions in a pregnant woman, usually around the time of delivery. It is the late complication of pre-eclamptic toxaemia (gestosis). Although its frequency in nations with excellent obstetric services has fallen below 1/500 pregnancies, it is still common in many other countries. The primary pathology is in the placenta, which secretes an anti-angiogenic factor in response to ischaemia, leading to endothelial dysfunction.[41][42] In fatal cases, there are arterial lesions in many organs including the brain. This is the second most frequent organic psychosis, and the second to be described.[43] Psychoses occur in about 5% of cases, and about 240 detailed cases have been reported.[30]: 28–35 It particularly affects first time mothers. Seizures may begin before, during or after labour, but the onset of psychosis is almost always postpartum. These mothers usually experience delirium but some have manic features. The duration is remarkably short, with a median duration of 8 days. This, together with the absence of a family history and of recurrences, contrasts with puerperal bipolar/cycloid psychoses. After recovery, amnesia and sometimes retrograde memory loss may occur, as well as other permanent cerebral lesions such as dysphasia, hemiplegia or blindness.
A variant was described by Donkin.[44] He had been trained by Simpson (one of those who first recognized the importance of albuminuria) in Edinburgh, and recognized that some cases of eclamptic psychosis occurred without seizures; this explains the interval between seizures (or coma) and psychosis, a gap that has occasionally exceeded 4 days: seizures and psychosis are two different consequences of severe gestosis. Donkin psychosis may not be rare: a British series included 13 possible cases;[30]: 35 but clarifying its distinction from postpartum bipolar disorder requires prospective investigations in collaboration with obstetricians.
Wernicke–Korsakoff psychosis[edit | edit source]
Wernicke–Korsakoff psychosis was described by Wernicke[45] and Korsakoff.[46] The pathology is damage to the core of the brain including the thalamus and mamillary bodies. Its most striking clinical feature is loss of memory, which can be permanent. It is usually found in severe alcoholics, but can also result from pernicious vomiting of pregnancy (hyperemesis gravidarum), because the requirement for thiamine is much increased in pregnancy; nearly 200 cases have been reported.[30]: 36–41 The cause is vitamin B1 (thiamine) deficiency. This has been available for treatment and prevention since 1936,[47] so the occurrence of this syndrome in pregnancy should be extinct. But these cases continue to be reported – more than 50 in this century – from all over the world, including some from countries with advanced medical services;[48] most are due to rehydration without vitamin supplements. A pregnant woman who presents in a dehydrated state due to pernicious vomiting urgently needs thiamine, as well as intravenous fluids.
Vascular disorders[edit | edit source]
Various vascular disorders occasionally cause psychosis, especially cerebral venous thrombosis. Puerperal women are liable to thrombosis, especially thrombophlebitis of the leg and pelvic veins; aseptic thrombi can also form in the dural venous sinuses and the cerebral veins draining into them. Most patients present with headache, vomiting, seizures and focal signs such as hemiplegia or dysphasia, but a minority of cases have a psychiatric presentation.[49] The incidence is about 1 in 1,000 births in Europe and North America,[50] but much higher in India, where large series have been collected.[51] Psychosis is occasionally associated with other arterial or venous lesions: epidural anaesthesia can, if the dura is punctured, lead to leakage of cerebrospinal fluid and subdural haematoma.[52] Arterial occlusion may be due to thrombi, amniotic fragments or air embolism. Postpartum cerebral angiopathy is a transitory arterial spasm of medium caliber cerebral arteries; it was first described in cocaine and amphetamine addicts, but can also complicate ergot and bromocriptine prescribed to inhibit lactation. Subarachnoid haemorrhage can occur after miscarriage or childbirth. All these usually present with neurological symptoms, and occasionally with delirium.
Epilepsy[edit | edit source]
Women with a lifelong epileptic history are liable to psychoses during pregnancy, labour and the puerperium. Women occasionally develop epilepsy for the first time in relation to their first pregnancy, and psychotic episodes have been described. There are over 30 cases in the literature.[30]: 56–58
Hypopituitarism[edit | edit source]
Pituitary necrosis following postpartum haemorrhage (Sheehan's syndrome) leads to failure and atrophy of the gonads, adrenal and thyroid. Chronic psychoses can supervene many years later, based on myxoedema, hypoglycaemia or Addisonian crisis. But these patients can also develop acute and recurrent psychoses, even as early as the puerperium.[53][30]: 59–62
Water intoxication[edit | edit source]
Hyponatraemia (which leads to delirium) can complicate oxytocin treatment, usually when given to induce an abortion. By 1975, 29 cases had been reported, of which three were severe or fatal.[54]
Urea cycle disorders[edit | edit source]
Inborn errors of the Krebs-Henseleit urea cycle lead to hyperammonaemia. In carriers and heterozygotes, encephalopathy can develop in pregnancy or the puerperium. Cases have been described in carbamoyl phosphate synthetase 1, argino-succinate synthetase and ornithine carbamoyltransferase deficiency.[30]: 63–65
Anti-NMDA receptor encephalitis[edit | edit source]
Anti-NMDA receptor encephalitis is an organic childbearing psychosis to be described is encephalitis associated with antibodies to the NMDA receptor; these women often have ovarian teratomas. A Japanese review found ten reported during pregnancy and five after delivery.[55]
Other organic with link to childbearing[edit | edit source]
Sydenham's chorea, of which chorea gravidarum is a severe variant, has a number of psychiatric complications, which include psychosis. This usually develops during pregnancy, and occasionally after the birth or abortion. Its symptoms include severe hypnagogic hallucinations (hypnagogia),[56][57] possibly the result of the extreme sleep disorder. This form of chorea was caused by streptococcal infections, which at present respond to antibiotics; it still occurs as a result of systemic lupus or anti-phospholipid syndromes. Only about 50 chorea psychoses have been reported, and only one this century; but it could return if the streptococcus escapes control. Alcohol withdrawal states (delirium tremens) occur in addicts whose intake has been interrupted by trauma or surgery; this can happen after childbirth. Postpartum confusional states have also been reported during withdrawal from opium[58] and barbiturates.[59] One would expect acquired immunodeficiency syndrome (HIV/AIDS) encephalitis to present in pregnancy or the puerperium, because it is a venereal disease that can progress rapidly; one case of AIDS encephalitis, presenting in the 28th week of gestation, has been reported from Haiti,[60] and there may be others in countries where AIDS is rife. Anaemia is common in pregnancy and the puerperium, and folate deficiency has been linked to psychosis.[61]
Incidental organic psychoses[edit | edit source]
The psychoses, mentioned above, all had a recognized connection with childbearing. But medical disorders with no specific link have presented with psychotic symptoms in the puerperium; in them the association seems to be fortuitous.[30]: 6–73 They include neurosyphilis, encephalitis including von Economo's, meningitis, cerebral tumours, thyroid disease and ischaemic heart disease.
Treatment[edit | edit source]
While each case is considered by individual circumstances, generally, PPP is assessed as a psychiatric emergency and requires admission to a psychiatric hospital for close care.[4][2] This admission may be to a mother-baby psychiatric unit or a general adult psychiatric unit, depending upon the availability in one's country and area.[3][2] Because few units, especially in the United States, offer mother-baby services, patients may be discharged home as soon as the worst concerns for the safety of the patient and infant are cleared, even though the patient may still be experiencing some symptoms of PPP; in this way, patients receive care to prevent harm to self and others but return home to care for their children and promote bonding as soon as they are able.[3][2]
Treatment plans are made up of a combination of education, medication, and close follow-up care and support;[3][2] the major goals of care include improving sleep and psychotic symptoms while helping to minimize major shifts in mood, such as depression and mania.[2] Medical treatment typically involves ECT, benzodiazepines, lithium, and/or antipsychotics.[3][4][2]
Lithium[edit | edit source]
Lithium is a psychiatric medication commonly classified as a "mood stabilizer" and often used for the treatment of bipolar disorder.[62] For PPP, lithium can be administered as a medication by itself ("monotherapy") or as an additional medication with other psychiatric drugs ("adjunctive therapy").[2] Research has shown strong evidence for the effectiveness of lithium as monotherapy in preventing repeat episodes of psychosis, particularly when compared to antipsychotic use alone,[3][2][9] and current recommendations suggest it as a first-line treatment for PPP in patients for whom this is safe[3][4][2] (lithium is not advised for patients with severe kidney or heart disease, thyroid dysfunction, Brugada syndrome, or who have known allergies to the drug).[63] Lithium has been associated with side effects to the fetus when taken during pregnancy, including body and heart abnormalities (e.g., Ebstein's anomaly); these effects have been documented in all trimesters, but higher risks, particularly for structural heart problems and spontaneous abortion, are typically seen with exposure in the first trimester.[63][64]
Lithium is also currently the recommended medication for prevention of psychotic episodes in individuals who have a known history of bipolar disorder and/or previous episodes of PPP.[4][2] The preferred strategy for preventative medication is to begin lithium immediately following delivery of the infant, minimizing the exposure of the fetus to lithium while in the womb.[2] Treatment throughout pregnancy, however, may be warranted as appropriate for treatment of bipolar disorder,[13] and is dosed according to appropriate medical advisory for the patient's trimester.[10] Lithium effectiveness is based on reaching an optimum level in the individual's blood, which usually requires more frequent bloodwork and adjustment of medication dosage to find and maintain an appropriate level.[63] Stopping lithium requires slow and gradual discontinuation; sudden removal of the medication may lead to symptom relapse and suicidal thoughts.[63]
Benzodiazepines[edit | edit source]
Benzodiazepines are a class of drug commonly used for anxiety disorders and insomnia, though they also play a role as an additive medication for psychosis and delirium.[65] According to a small study of 64 women, benzodiazepines had a minor but positive effect in reducing psychosis as a sequentially added medication for treatment of PPP (on top of lithium and antipsychotic medications);[66] due to little supporting data, benzodiazepines are currently only recommended as an add-on medication to lithium and/or antipsychotic drugs, usually in the setting of continued sleep disturbance despite the use of the other medications.[4][2]

Antipsychotics[edit | edit source]
Antipsychotic medications are the preferred class of drug used to treat general psychosis, including schizophrenia spectrum disorders.[65] Few studies provide evidence for the efficacy of antipsychotic use in PPP, with the exception of one study of 64 women which examined antipsychotic use as a drug by itself and combined with lithium.[66] In this study, antipsychotic use was effective in both scenarios, but not as effective as lithium used by itself.[66] Despite this limited evidence in PPP specifically, current recommendations include antipsychotic use as an additional medication for patients receiving lithium, or antipsychotic use on its own for those who are unable to tolerate lithium.[2]
Antipsychotic choice is physician- and case-dependent, with a large number of available medications offering a range of nuanced effects. Treatment guidance is largely based on recommendations and data from patients with schizophrenia who become pregnant.[67] First-generation antipsychotics have a longer history of use, efficacy and safety in pregnancy, particularly chlorpromazine and haloperidol.[67] Still, second-generation antipsychotics may be preferred over first-generation antipsychotics due to the reduced risk for extrapyramidal symptoms (e.g., uncontrollable movements, tremors or muscle contractions).[68]
Electroconvulsive therapy[edit | edit source]

Electroconvulsive therapy (ECT) has a long and well-documented history as a psychiatric treatment, starting in 1938, with a large body of literature supporting its effectiveness and safety in various psychiatric conditions, including psychosis.[69][70] However, while it has been used for PPP for over 50 years, the data on its efficacy in PPP specifically is limited to a significant number of case series and various retrospective matched-cohort studies.[69] There are few adverse effects associated with ECT, though short-term memory disturbance (< 6 months) has been reported.[70] Patients who are adverse to long-term psychiatric medications may prefer ECT, particularly given the minimal and short-term impact that ECT-related medications (e.g., drugs for muscle relaxation and anesthesia) appear to have on breastfeeding (as compared to psychiatric medications).[69] Per current recommendations, ECT is considered an option for PPP when medications are ineffective.[2]
Breastfeeding[edit | edit source]
Disruption of continued contact with the infant may occur during hospitalization and treatment, which may impact breastfeeding capacity;[2] hospital units may provide the use of a breast pump to mitigate this concern.[2] Strategies and concerns should also be discussed regarding breastfeeding and its impact on sleep, as poor sleep related to night feedings may worsen or delay patient recovery; alternatives may include social support and bottle-feeding through the night to allow the patient time for adequate rest.[2]
The effects of various medications during breastfeeding are poorly studied.[67] According to one systematic review evaluating 37 different reports of antipsychotic use in 206 infants, olanzapine has the strongest supporting evidence for low infant exposure through breastmilk, while fewer reports support a similarly low exposure for quetiapine and ziprasidone use.[71] Chlorpromazine also demonstrates minimal transference to the infant through breastmilk.[72]
There is currently no consensus to the safety or level of lithium present in breastmilk, though several guidelines and reviews do not consider it an absolute danger to the infant which should exclude its use.[73] Several reports describe safe use of lithium during breastfeeding with no noticeable effects in the infant, though any lasting effects have not been well-studied.[73] As in adults, lithium use with breastfeeding infants requires careful monitoring of the child for serum levels, dose adjustment, and any side effects; thus, the ability of the patient to maintain follow-up care should also be taken into consideration when lithium may be used.[73]
Short-acting benzodiazepines, like lorazepam, are preferred from this class of drugs as they demonstrate lower levels passed through breastmilk and no reported side effects in infants.[74][75]
Outcomes[edit | edit source]
Symptoms may last for a variable length of time (up to one year in 25% of cases) despite adequate treatment.[2] Despite most individuals (50-80%) experiencing a relapse episode and development of chronic psychiatric disorders (such as bipolar spectrum disorder), these same individuals are expected to be able to resume normal activities of their daily life with the same level of function as previously experienced.[2][76] Of the minority of persons who experience PPP and choose to have another pregnancy, evidence has shown that about 33% will have a repeat psychotic episode.[2][13] Other factors that contribute to poorer prognosis include an experience of PPP that's limited to the postpartum period (rather than a diagnosis of bipolar disorder, for example), longer initial psychotic episodes, and a higher severity in the initial episode.[2][13]
Infanticide (or filicide) is thought to occur in 1 to 4% of PPP cases,[3][4][2][77] and some evidence suggests that these incidents are more commonly related to PPP episodes that feature more depressive symptoms.[77]
Epidemiology[edit | edit source]
PPP is rare, reported to occur in about 1 to 2 of every 1000 childbirths (0.9 to 2.6 per 1,000).[2][78] Reported cases are thought to underestimate the actual occurrence of PPP due to the probability of some individuals avoiding hospitalized care to avoid separation from their child (particularly in locations with no mother-baby units) or fear of stigma, as well as the likelihood of misdiagnosis with other postpartum disorders.[10] The first month following childbirth is associated with a higher relative risk for hospital admission due to psychosis when compared to other times in an individual's life.[79] While no specific genetic factors have been linked to PPP, a family history or personal history of bipolar disorder has been strongly associated with higher risk for PPP episodes.[2]
History[edit | edit source]
Postpartum psychosis had been recognized in earlier editions of the DSM (I and II), first as Involutional Psychotic Reaction and later as Psychosis with Childbirth.[80] It was removed in the DSMIII following arguments that psychiatric disorders associated with pregnancy and childbirth were no different than other psychiatric illnesses; it has only been more recently recognized again with the 1994 release of the DSM-IV, when the specifier "with postpartum onset" was included for various diagnoses.[80]
Between the 16th and 18th centuries, about 50 brief reports regarding postpartum psychosis were published; among them is the observation that these psychoses could recur,[81] and that they occur both in breast-feeding and non-lactating women.[82] In 1797, Osiander, an obstetrician from Tübingen, reported two cases at length that contributed significantly to the knowledge of this disorder during that time.[83] In 1819, Esquirol conducted a survey of cases admitted to the Salpêtrière, and pioneered long-term studies.[84] From that time, puerperal psychosis became widely known to the medical profession. In the next 200 years, over 2,500 theses, articles and books were published. Among these many contributions were Delay's unique investigation using serial curettage [85] and Kendell's record-linkage study comparing 2 years before and 2 years after the birth.[86] In the last few years, two monographs reviewed over 2,400 works, detailing more than 4,000 cases of childbearing psychoses from the literature and a personal series of more than 320 cases.[87][30]
Society and culture[edit | edit source]
Support[edit | edit source]
In the UK, a series of workshops called "Unravelling Eve" were held in 2011, where women who had experienced postpartum depression shared their stories.[88]
Cases[edit | edit source]
Harriet Sarah, Lady Mordaunt (1848–1906), formerly Harriet Moncreiffe, was the Scottish wife of an English baronet and Member of Parliament, Sir Charles Mordaunt.[citation needed] She was the defendant in a divorce case in which the Prince of Wales (later King Edward VII) was involved; after a trial lasting seven days, the jury determined that Lady Mordaunt had "puerperal mania" and her husband's petition for divorce was dismissed, while Lady Mordaunt was committed to an asylum.[89]
Andrea Yates had depression and, four months after the birth of her fifth child, relapsed, with psychotic features. Several weeks later she drowned all five children. Under the law in Texas, she was sentenced to life imprisonment, but, after a retrial, was committed to a mental hospital.
Lindsay Clancy[edit | edit source]
Lindsay Marie Clancy (née Musgrove;[90] born August 11, 1990) is an American from Duxbury, Massachusetts who allegedly strangled her three children on the evening of January 24, 2023.[91][92] Two of the children, 5-year-old Cora Clancy and 3-year-old Dawson Clancy, were pronounced dead at the hospital on January 24, 2023, while a third child, 7-month-old Callan Clancy (who turned 8-months old while hospitalized), died on January 27, 2023. After she strangled the children she attempted to kill herself by jumping from a window. She is now paralyzed from the waist down.[93] Experts for the defense believe Clancy had postpartum psychosis[94] and that she may have been "overmedicated."[95] Her attorneys reported that, in the 4 months leading up to the incident, Lindsay had been prescribed 13 different psychiatric medications.[91]
Clancy worked as a labor and delivery nurse at Massachusetts General Hospital. She also made videos for newborn mothers.[96] Prior to the incident, she struggled with various facets of her mental health, seeking treatment from one psychiatrist and a psychiatric nurse practitioner. She was admitted to McLean Hospital less than three weeks before the killings.[91][96]
The incident ignited a discussion on social media platforms such as TikTok about postpartum psychosis and various others struggles mothers face after a birth.[97] This had led to many mothers speaking out about their own stories, opening up on social media or to various press outlets about their experiences.[98] Several bills were introduced to the Massachusetts state legislature in an attempt to better address postpartum illnesses in the state.[99]
In fiction[edit | edit source]
Guy de Maupassant, in his novel Mont-Oriol (1887) described a brief postpartum psychotic episode.
Charlotte Perkins Gilman, in her short story The Yellow Wallpaper (1892) described severe depression with psychotic features starting after childbirth, perhaps similar to that experienced by the author herself.
Stacey Slater, a fictional character in the long-running BBC soap-opera EastEnders had postpartum psychosis in 2016, and was one of the show's biggest storylines that year.[citation needed]
In the House, M.D. episode Forever, House takes the case of a mother with postpartum psychosis.
Legal status[edit | edit source]
Postpartum psychosis, especially when there is a marked component of depression, has a small risk of filicide. In acute manic or cycloid cases, this risk is about 1%.[30]: 240–246 Most of these incidents have occurred before the mother came under treatment, and some have been accidental. Several nations including Canada, United Kingdom, Australia, and Italy recognize postpartum mental illness as a mitigating factor in cases where mothers kill their children.[100] In the United States, such a legal distinction was not made as of 2009,[100] and an insanity defense is not available in all states.[101]
The United Kingdom has had the Infanticide Act since 1922.
Books[edit | edit source]
The following books have been published about these psychoses:
- Ripping, Dr (1877) Die Geistesstörungen der Schwangeren, Wöchnerinnen und Säugenden. Stuttgart, Enke.
- Knauer O (1897) Über Puerperale Psychose für practische Ärzte. Berlin, Karger.
- Twomey T (2009) Understanding Postpartum Psychosis: A Temporary Madness. Westport, Praeger.
- Harwood D (2017) Birth of a New Brain - Healing from Postpartum Bipolar Disorder. Brentwood, Post Hill Press.
References[edit | edit source]
- ↑ 1.0 1.1 1.2 1.3 "Postpartum psychosis". 11 February 2021. Archived from the original on 7 March 2021. Retrieved 6 January 2024.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 2.17 2.18 2.19 2.20 2.21 2.22 2.23 2.24 2.25 2.26 2.27 2.28 2.29 2.30 2.31 2.32 2.33 2.34 2.35 2.36 2.37 2.38 2.39 2.40 2.41 2.42 2.43 2.44 2.45 2.46 2.47 2.48 2.49 2.50 2.51 2.52 2.53 2.54 2.55 2.56 2.57 2.58 2.59 2.60 2.61 2.62 2.63 2.64 2.65 Hutner LA, Catapano LA, Nagle-Yang SM, Williams KE, Osborne LM, eds. (2021). Textbook of Women's Reproductive Mental Health. Washington, D.C.: American Psychiatric Association Publishing. ISBN 978-1-61537-386-4. OCLC 1289371393. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 3.17 3.18 3.19 3.20 3.21 3.22 3.23 3.24 3.25 3.26 3.27 3.28 3.29 Osborne LM (September 2018). "Recognizing and Managing Postpartum Psychosis: A Clinical Guide for Obstetric Providers". Obstetrics and Gynecology Clinics of North America. 45 (3): 455–468. doi:10.1016/j.ogc.2018.04.005. PMC 6174883. PMID 30092921.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 4.18 4.19 4.20 4.21 4.22 4.23 4.24 4.25 4.26 4.27 4.28 Rodriguez-Cabezas L, Clark C (September 2018). "Psychiatric Emergencies in Pregnancy and Postpartum". Clinical Obstetrics and Gynecology. 61 (3): 615–627. doi:10.1097/GRF.0000000000000377. PMC 6143388. PMID 29794819.
- ↑ Diagnostic and statistical manual of mental disorders: DSM-5-TR™ (Fifth, text revision ed.). Washington, DC: American Psychiatric Association Publishing. 2022. p. 108. ISBN 9780890425763.
- ↑ 6.0 6.1 6.2 Lewis G, Blake L, Seneviratne G (September 2022). "Delusional Misidentification Syndromes in Postpartum Psychosis: A Systematic Review". Psychopathology. 56 (4): 285–294. doi:10.1159/000526129. PMID 36116435. S2CID 252341410.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 American Psychiatric Association. DSM-5 Task Force (2017). Diagnostic and statistical manual of mental disorders: DSM-5 (Fifth ed.). New Delhi: American Psychiatric Association Publishing. ISBN 978-93-86217-96-7. OCLC 1030754444. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 8.0 8.1 8.2 Perry A, Gordon-Smith K, Jones L, Jones I (January 2021). "Phenomenology, Epidemiology and Aetiology of Postpartum Psychosis: A Review". Brain Sciences. 11 (1): 47. doi:10.3390/brainsci11010047. PMC 7824357. PMID 33406713.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Bergink V, Rasgon N, Wisner KL (December 2016). "Postpartum Psychosis: Madness, Mania, and Melancholia in Motherhood". The American Journal of Psychiatry. 173 (12): 1179–1188. doi:10.1176/appi.ajp.2016.16040454. PMID 27609245. S2CID 27115535.
- ↑ 10.0 10.1 10.2 Jones I, Chandra PS, Dazzan P, Howard LM (November 2014). "Bipolar disorder, affective psychosis, and schizophrenia in pregnancy and the post-partum period". Lancet. 384 (9956): 1789–1799. doi:10.1016/S0140-6736(14)61278-2. PMID 25455249. S2CID 44481055.
- ↑ Kaplan DA, Greene JA (2020-05-01). "Postpartum Obsessive-Compulsive Disorder". In Robinson GE, Nadelson CC, Apter G (eds.). Postpartum Mental Health Disorders: A Casebook. pp. 51–60. doi:10.1093/med/9780190849955.003.0007. ISBN 978-0-19-084995-5. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ Kamperman AM, Veldman-Hoek MJ, Wesseloo R, Robertson Blackmore E, Bergink V (September 2017). "Phenotypical characteristics of postpartum psychosis: A clinical cohort study". Bipolar Disorders. 19 (6): 450–457. doi:10.1111/bdi.12523. PMID 28699248. S2CID 32029514.
- ↑ 13.0 13.1 13.2 13.3 Wesseloo R, Kamperman AM, Munk-Olsen T, Pop VJ, Kushner SA, Bergink V (February 2016). "Risk of Postpartum Relapse in Bipolar Disorder and Postpartum Psychosis: A Systematic Review and Meta-Analysis". The American Journal of Psychiatry. 173 (2): 117–127. doi:10.1176/appi.ajp.2015.15010124. PMID 26514657.
- ↑ Nguyen K, Mukona LT, Nalbandyan L, Yar N, St Fleur G, Mukona L, et al. (September 2022). "Peripartum Complications as Risk Factors for Postpartum Psychosis: A Systemic Review". Cureus. 14 (9): e29224. doi:10.7759/cureus.29224. PMC 9495292. PMID 36159350.
- ↑ Glover L, Jomeen J, Urquhart T, Martin CR (2014-05-27). "Puerperal psychosis – a qualitative study of women's experiences". Journal of Reproductive and Infant Psychology. 32 (3): 254–269. doi:10.1080/02646838.2014.883597. ISSN 0264-6838. S2CID 144220513. Archived from the original on 2023-05-07. Retrieved 2024-01-06.
- ↑ Coyle N, Jones I, Robertson E, Lendon C, Craddock N (October 2000). "Variation at the serotonin transporter gene influences susceptibility to bipolar affective puerperal psychosis". Lancet. 356 (9240): 1490–1491. doi:10.1016/S0140-6736(00)02877-4. PMID 11081536. S2CID 6095129.
- ↑ Jones I, Hamshere M, Nangle JM, Bennett P, Green E, Heron J, et al. (July 2007). "Bipolar affective puerperal psychosis: genome-wide significant evidence for linkage to chromosome 16". The American Journal of Psychiatry. 164 (7): 1099–1104. doi:10.1176/ajp.2007.164.7.1099. PMID 17606662.
- ↑ Kumar HB, Purushottam M, Kubendran S, Gayathri P, Mukherjee O, Murthy AR, et al. (October 2007). "Serotonergic candidate genes and puerperal psychosis: an association study". Psychiatric Genetics. 17 (5): 253–260. doi:10.1097/YPG.0b013e3280ae6cc3. PMID 17728663. S2CID 38216613.
- ↑ Thippeswamy H, Paul P, Purushottam M, Philip M, Jain S, Chandra PS (April 2017). "Estrogen pathway related genes and their association with risk of postpartum psychosis: A case control study". Asian Journal of Psychiatry. 26: 82–85. doi:10.1016/j.ajp.2017.01.014. PMID 28483099.
- ↑ Schiller CE, Meltzer-Brody S, Rubinow DR (February 2015). "The role of reproductive hormones in postpartum depression". CNS Spectrums. 20 (1): 48–59. doi:10.1017/S1092852914000480. PMC 4363269. PMID 25263255.
- ↑ Del Río JP, Alliende MI, Molina N, Serrano FG, Molina S, Vigil P (2018). "Steroid Hormones and Their Action in Women's Brains: The Importance of Hormonal Balance". Frontiers in Public Health. 6: 141. doi:10.3389/fpubh.2018.00141. PMC 5974145. PMID 29876339.
- ↑ Stahl SM, Grady MM, Muntner N (2021). Stahl's essential psychopharmacology: neuroscientific basis and practical applications (Fifth ed.). Cambridge, United Kingdom. ISBN 978-1-108-97529-2. OCLC 1253443384. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 23.0 23.1 23.2 Dye C, Lenz KM, Leuner B (2021). "Immune System Alterations and Postpartum Mental Illness: Evidence From Basic and Clinical Research". Frontiers in Global Women's Health. 2: 758748. doi:10.3389/fgwh.2021.758748. PMC 8866762. PMID 35224544.
- ↑ Dazzan P, Fusté M, Davies W (November 2018). "Do Defective Immune System-Mediated Myelination Processes Increase Postpartum Psychosis Risk?". Trends in Molecular Medicine. 24 (11): 942–949. doi:10.1016/j.molmed.2018.09.002. PMC 6224363. PMID 30348609.
- ↑ Lewis KJ, Di Florio A, Forty L, Gordon-Smith K, Perry A, Craddock N, et al. (January 2018). "Mania triggered by sleep loss and risk of postpartum psychosis in women with bipolar disorder" (PDF). Journal of Affective Disorders. 225: 624–629. doi:10.1016/j.jad.2017.08.054. PMID 28889048. S2CID 205644290. Archived (PDF) from the original on 2023-06-01. Retrieved 2024-01-06.
- ↑ Strömgren E (1986) Reactive (psychogenic) psychoses and their relations to schizoaffective psychoses. In A Marneros & M T Tsuang (editors), Schizoaffective Psychoses, Berlin-Heidelberg, Springer-Verlag, pages 260-271.
- ↑ Edelberg H, Galant (1925) Über psychotische Zustände nach künstlichen Abort. Zeitschrift für die gesamte Neurologie und Psychiatrie 97: 106-128.
- ↑ Trixler M, Jádi T, Wagner M (1981) Adoptació utáni ‚post partum' pszichózisok. Orvosi Hetilap 122: 3071-3071.
- ↑ Ginath Y (1974) Psychoses in males in relation to their wives' pregnancy and childbirth. Israeli Annals of Psychiatry and Related Disciplines 12: 227-237.
- ↑ 30.00 30.01 30.02 30.03 30.04 30.05 30.06 30.07 30.08 30.09 30.10 30.11 30.12 30.13 Brockington IF (2017). The Psychoses of Menstruation and Childbearing. Cambridge: Cambridge University Press. pp. 1–278.
- ↑ Yamada N, Fukui M, Ishii K, Shibata H, Okabe H, Ohomiya H, Matsunobu A, Nishizima M 1980) A case of adult form hypercitrullinemia with consciousness disturbance and marked hypertransaminasenemia after delivery. Nihon Shokakibyo Gakkae Zasshi 77: 1655-1660.
- ↑ Ito Y, Abe T, Tomioka R, Komori T, Araki N (2010) [Anti-NMDA receptor encephalitis during pregnancy]. Rinsho Shinkeigaku 50: 103-107.
- ↑ Ndosi N K, Mtawali M L (2002) The nature of puerperal psychosis at Muhimbili National Hospital: its physical co-morbidity, associated main obstetric and social factors. African Journal of Reproductive Health 6: 41-49.
- ↑ Hippocrates (5th Century BC) Epidemics, book III, volume 1, pages 280-283, in the edition translated by W H S Jones, 1931.
- ↑ Chaslin P (1895) Confusion Mentale Primitive, Stupidité, Démence aiguë, Stupeur Primitive. Paris, Harmattan.
- ↑ Bonhöffer K (1910) Die symptomatischen Psychosen im Gefolge von akuten Infektionen und inneren Erkrankungen. Leipzig and Wien, Deutlicke.
- ↑ Poulsen A (1899) Nogle bemärkungen om puerperal psychosen. Hospitals-Tidende, 4 räkke, 7: 251-260.
- ↑ Halim A, Utz B, Biswas A, Rahman F, van den Broek N (2014) Cause of, and contributing factors, to maternal deaths: a cross-sectional study using verbal autopsy in four districts in Bangladesh. British Journal of Obstetrics & Gynaecology 121, supplement s4: 86-94.
- ↑ Guerrier G, Oluyide B, Keramarou M, Grais R (2013) High maternal and neonatal mortality rates in northern Nigeria: an 8-month observational study. International Journal of Women's Health 5: 495-499.
- ↑ Vallely L, Ahmed Y, Murray S F (2005) Postpartum maternal morbidity requiring hospital admission in Lusaka, Zambia – a descriptive study. BMC Pregnancy Childbirth 5: 1-10.
- ↑ Roberts J M (1998) Endothelial dysfunction in pre-eclampsia. Seminars in Reproductive Endocrinology 16: 5-15.
- ↑ Tomimatsu T, Mimura K, Matsuzaki S, Endo M, Kumasawa K, Kimura T (2019) Preeeclampsia: maternal systemic vascular disorder caused by generalized endothelial dysfunction due to placental antiangiogenic factors. International Journal of Molecular Science 20: E4246.
- ↑ Imbert-Gourbeyer (1861) Des paralyses puerpérales. Mémoirs de l'Académie Impériale de Médecine 25: 46-53.
- ↑ Donkin A S (1863) On the pathological relation between albuminuria and puerperal mania. Edinburgh Medical Journal 8: 994-1004.
- ↑ Wernicke C (1881) Lehrbuch der Gehirnkrankheiten für Ärzte und Studirende, volume 2. Kassel & Berlin, Fischer, pages 229-242.
- ↑ Korsakow S S (1887) Über eine besonderer Form psychischer Störung. Archiv für Psychiatrie 21: 671-704.
- ↑ Williams R R, Cline J K (1936) Synthesis of vitamin B1. Journal of the American Chemical Association 58: 1504-1505.
- ↑ Giugale L E, Young O M, Streitman D C (2015) Iatrogenic Wernicke encephalopathy in a patient with severe hyperemesis gravidarum. Obstetrics and Gynecology 125: 1150-1152.
- ↑ Kalbag R M, Woolf A L (1967) Cerebral Venous Thrombosis, with Special Reference to Primary Aseptic Thrombosis. Oxford, Oxford University Press.
- ↑ Lanska D J, Kryscio R J (2000) Risk factors for peripartum and postpartum stroke and intracranial venous thrombosis. Stroke 31: 1274-1282.
- ↑ Srinavasan K (1988) Puerperal cerebral venous and arterial thrombosis. Seminars in Neurology 8:222-225.
- ↑ Jack T M (1982) Post-partum intracranial subdural haematoma. A possible complication of epidural analgesia. British Medical Journal 285: 972 only.
- ↑ Shoib S, Dar M M, Arif T, Bashir H, Bhat M H, Ahmed J (2013) Sheehan's syndrome presenting as psychosis: a rare clinical presentation. Medical Journal of the Republic of Iran 27: 35-37.
- ↑ Ahmad A J, Clark E H, Jacobs H S (1975) Water intoxication associated with oxytocin infusion. Postgraduate Medical Journal 51: 249-252.
- ↑ Doden T, Sekijima Y, Ikeda J, Ozawa K, Ohashi N, Kodaira M, Hineno A, Tachibana N, Ikeda S (2016) Postpartum anti-N-methyl-D-aspartate receptor encephalitis: a case report and literature review. Internal Medicine 56: 357-362.
- ↑ Marcé L V (1860) L'État mental dans la chorée. Mémoirés de l'Académie de Médecine 24: 30-38.
- ↑ Breton A (1893) État mental dans la chorée. Thèse, Paris, no. 124.
- ↑ Hill N M (1891) Four cases of puerperal insanity. Transactions of the Iowa State Medical Society 9: 132-134.
- ↑ Baker A A (1967) Psychiatric Disorders in Obstetrics, Oxford, Blackwell, page 61.
- ↑ Birnbach D J, Bourlier R A, Choi R, Thys D M (1995) Anaesthetic management of Caesarean section in a patient with recurrent genital herpes and AIDS-related dementia. British Journal of Anaesthesia 75: 639-641.
- ↑ Thornton W E (1977) Folate deficiency in puerperal psychosis. American Journal of Obstetrics and Gynecology 129: 222-223.
- ↑ Stahl, Stephen M. (2021). Stahl's essential psychopharmacology: neuroscientific basis and practical applications. Meghan M. Grady, Nancy Muntner (Fifth ed.). Cambridge, United Kingdom. ISBN 978-1-108-97529-2. OCLC 1253443384. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 63.0 63.1 63.2 63.3 Stahl, Stephen M., ed. (2020), "Lithium", Prescriber's Guide: Stahl's Essential Psychopharmacology (7 ed.), Cambridge: Cambridge University Press, pp. 415–420, doi:10.1017/9781108921275.071, ISBN 978-1-108-92601-0, archived from the original on 2024-01-11, retrieved 2023-01-23
- ↑ Fornaro, Michele; Maritan, Elena; Ferranti, Roberta; Zaninotto, Leonardo; Miola, Alessandro; Anastasia, Annalisa; Murru, Andrea; Solé, Eva; Stubbs, Brendon; Carvalho, André F.; Serretti, Alessandro; Vieta, Eduard; Fusar-Poli, Paolo; McGuire, Philip; Young, Allan H. (2020-01-01). "Lithium Exposure During Pregnancy and the Postpartum Period: A Systematic Review and Meta-Analysis of Safety and Efficacy Outcomes". American Journal of Psychiatry. 177 (1): 76–92. doi:10.1176/appi.ajp.2019.19030228. hdl:2445/148139. ISSN 0002-953X. PMID 31623458. S2CID 202253605. Archived from the original on 2023-01-23. Retrieved 2024-01-06.
- ↑ 65.0 65.1 Stahl, Stephen M. (2020-11-19). Prescriber's Guide: Stahl's Essential Psychopharmacology (7 ed.). Cambridge University Press. doi:10.1017/9781108921275.075. ISBN 978-1-108-92127-5. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 66.0 66.1 66.2 Bergink, Veerle; Burgerhout, Karin M.; Koorengevel, Kathelijne M.; Kamperman, Astrid M.; Hoogendijk, Witte J.; Lambregtse-van den Berg, Mijke P.; Kushner, Steven A. (2015-02-01). "Treatment of psychosis and mania in the postpartum period". The American Journal of Psychiatry. 172 (2): 115–123. doi:10.1176/appi.ajp.2014.13121652. ISSN 1535-7228. PMID 25640930. Archived from the original on 2023-07-18. Retrieved 2024-01-06.
- ↑ 67.0 67.1 67.2 Smith, Beth; Dubovsky, Steven L. (2017-11-02). "Pharmacotherapy of mood disorders and psychosis in pre- and post-natal women". Expert Opinion on Pharmacotherapy. 18 (16): 1703–1719. doi:10.1080/14656566.2017.1391789. ISSN 1465-6566. PMID 29019422. S2CID 46850504. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ Leucht, Stefan; Corves, Caroline; Arbter, Dieter; Engel, Rolf R.; Li, Chunbo; Davis, John M. (2009-01-03). "Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis". Lancet. 373 (9657): 31–41. doi:10.1016/S0140-6736(08)61764-X. ISSN 1474-547X. PMID 19058842. S2CID 1071537. Archived from the original on 2023-08-25. Retrieved 2024-01-06.
- ↑ 69.0 69.1 69.2 Focht, Amanda; Kellner, Charles H. (March 2012). "Electroconvulsive Therapy (ECT) in the Treatment of Postpartum Psychosis". The Journal of ECT. 28 (1): 31–33. doi:10.1097/YCT.0b013e3182315aa8. ISSN 1095-0680. PMID 22330704. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 70.0 70.1 Gazdag, Gábor; Ungvari, Gabor S. (2019-01-04). "Electroconvulsive therapy: 80 years old and still going strong". World Journal of Psychiatry. 9 (1): 1–6. doi:10.5498/wjp.v9.i1.1. ISSN 2220-3206. PMC 6323557. PMID 30631748.
- ↑ Uguz, Faruk (June 2016). "Second-Generation Antipsychotics During the Lactation Period: A Comparative Systematic Review on Infant Safety". Journal of Clinical Psychopharmacology. 36 (3): 244–252. doi:10.1097/JCP.0000000000000491. ISSN 1533-712X. PMID 27028982. S2CID 38589732. Archived from the original on 2023-01-24. Retrieved 2024-01-06.
- ↑ Newton, Edward R.; Hale, Thomas W. (December 2015). "Drugs in Breast Milk". Clinical Obstetrics & Gynecology. 58 (4): 868–884. doi:10.1097/GRF.0000000000000142. ISSN 0009-9201. PMID 26457856. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ 73.0 73.1 73.2 "Lithium", Drugs and Lactation Database (LactMed®), Bethesda (MD): National Institute of Child Health and Human Development, 2006, PMID 30000212, archived from the original on 2023-03-24, retrieved 2023-01-24
- ↑ Raza, Sehar K.; Raza, Syed (2022), "Postpartum Psychosis", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 31335024, archived from the original on 2023-02-10, retrieved 2023-01-24
- ↑ "Lorazepam", Drugs and Lactation Database (LactMed®), Bethesda (MD): National Institute of Child Health and Human Development, 2006, PMID 30000290, archived from the original on 2022-10-13, retrieved 2023-01-24
- ↑ Gilden J, Kamperman AM, Munk-Olsen T, Hoogendijk WJ, Kushner SA, Bergink V (March 2020). "Long-Term Outcomes of Postpartum Psychosis: A Systematic Review and Meta-Analysis". The Journal of Clinical Psychiatry. 81 (2): 10750. doi:10.4088/JCP.19r12906. PMID 32160423. S2CID 212677987.
- ↑ 77.0 77.1 Brockington I (February 2017). "Suicide and filicide in postpartum psychosis". Archives of Women's Mental Health. 20 (1): 63–69. doi:10.1007/s00737-016-0675-8. PMC 5237439. PMID 27778148.
- ↑ VanderKruik, Rachel; Barreix, Maria; Chou, Doris; Allen, Tomas; Say, Lale; Cohen, Lee S.; Maternal Morbidity Working Group (2017-07-28). "The global prevalence of postpartum psychosis: a systematic review". BMC Psychiatry. 17 (1): 272. doi:10.1186/s12888-017-1427-7. ISSN 1471-244X. PMC 5534064. PMID 28754094.
- ↑ Tschinkel, S.; Harris, M.; Le Noury, J.; Healy, D. (April 2007). "Postpartum psychosis: two cohorts compared, 1875-1924 and 1994-2005". Psychological Medicine. 37 (4): 529–536. doi:10.1017/S0033291706009202. ISSN 0033-2917. PMID 17076918. S2CID 1820395. Archived from the original on 2023-01-24. Retrieved 2024-01-06.
- ↑ 80.0 80.1 Spinelli, Margaret (2021-10-01). "Postpartum psychosis: a diagnosis for the DSMV". Archives of Women's Mental Health. 24 (5): 817–822. doi:10.1007/s00737-021-01175-8. ISSN 1435-1102. PMID 34494144. S2CID 237437052. Archived from the original on 2024-01-11. Retrieved 2024-01-06.
- ↑ Van Foreest P (1609) Puerperas nonnunquam phreniticas fierit & sineglectim habeantur, sibi ipsis vim inferre. Observationes, scholio 7, lib. 10.
- ↑ Bartholomaeo de Battista a St Georgio (1784) Von der Tollsucht den Kindbetterinnen, in his Abhandlung von den Krankheiten des schönen Geschlechtes, Vienna, Sonnleithner, pages 113-114 in the 1819 edition.
- ↑ Osiander F B (1797). Neue Denkwürdigkeiten für Ärzte und Geburtshelfer. Göttingen, Rosenbusch, volume 1, pages 52-89, 90-128. These cases have been translated in reference 86, p19-30.
- ↑ Esquirol J E D (1819) De l'aliénation mentale des nouvelles accouchées et des nourrices. Annuaires Médicales-chirurgiques des Hôpitaux de Paris 1: 600-632.
- ↑ Delay J, Boitelle G, Corteel A (1948. Les psychoses du postpartum: étude cyto-hormonale. Semaines d'Hôpitaux de Paris 24: 2891-2901.
- ↑ Kendell RE, Chalmers JC, Platz C (May 1987). "Epidemiology of puerperal psychoses". The British Journal of Psychiatry. 150 (5): 662–73. doi:10.1192/bjp.150.5.662. PMID 3651704. S2CID 4613996.
- ↑ Brockington I F (2014) What is Worth Knowing about 'Puerperal Psychosis'. Bredenbury, Eyry Press. A pdf of this limited edition can be obtained gratis on email request to i.f.brockington@bham.ac.uk.
- ↑ Dolman C (4 December 2011). "When having a baby can cause you to 'lose your mind'". BBC News. Archived from the original on 7 September 2023. Retrieved 6 January 2024.
- ↑ Souhami D (1996). Mrs. Keppel and her daughter (1st U.S. ed.). New York: St. Martin's Press. ISBN 978-0312155940. E-book ISBN 9781466883505
- ↑ Backus, Lisa; Zaretsky, Mark (3 February 2023). "Attorney: Former CT resident Lindsay Clancy was 'overmedicated' when kids were killed". CT Insider. Archived from the original on 7 February 2023. Retrieved 7 February 2023.
- ↑ 91.0 91.1 91.2 Barry, Ellen (2023-02-08). "A Troubled Mother Faces Murder Charges in Her Young Children's Deaths". The New York Times. ISSN 0362-4331. Archived from the original on 2023-03-02. Retrieved 2023-03-02.
- ↑ WCVB Staff (4 February 2023). "Mother accused of killing her 3 kids before attempting suicide, making progress in hospital". ABC News. Archived from the original on 7 February 2023. Retrieved 7 February 2023.
- ↑ WCVB Staff (2 February 2023). "Lindsay Clancy, Duxbury mother accused of killing her 3 kids, making progress in hospital". ABC News. Archived from the original on 7 February 2023. Retrieved 7 February 2023.
- ↑ Wornell, Tyler (31 January 2023). "Postpartum psychosis in spotlight after death of three kids". NewsNation. Nexstar Media Inc. Archived from the original on 7 February 2023. Retrieved 7 February 2023.
- ↑ Carbonaro, Giulia (4 February 2023). "Lindsay Clancy to Face Charges as Attorney Points Out 'Overmedication'". Newsweek. NEWSWEEK DIGITAL LLC. Archived from the original on 7 February 2023. Retrieved 7 February 2023.
- ↑ 96.0 96.1 "Lindsay Clancy: A timeline of the Duxbury mother's mental health treatment". www.boston.com. Archived from the original on 2023-03-01. Retrieved 2023-03-02.
- ↑ Ryu, Jenna. "A mom is accused of killing her 3 kids. It sparked a national conversation about postpartum psychosis". USA TODAY. Archived from the original on 2023-03-02. Retrieved 2023-03-02.
- ↑ ""I wanted to put a knife into my baby": Moms on their postpartum psychosis". Newsweek. 2023-02-04. Archived from the original on 2023-03-02. Retrieved 2023-03-02.
- ↑ Aponte, Gabriella. "Postpartum depression focus for several Mass. bills after Clancy case". MetroWest Daily News. Archived from the original on 2023-03-02. Retrieved 2023-03-02.
- ↑ 100.0 100.1 Appel JM (8 November 2009). "When Infanticide Isn't Murder". The Huffington Post. Archived from the original on 12 March 2017. Retrieved 6 January 2024.
- ↑ Rhodes AM, Segre LS (August 2013). "Perinatal depression: a review of US legislation and law". Archives of Women's Mental Health. 16 (4): 259–270. doi:10.1007/s00737-013-0359-6. PMC 3725295. PMID 23740222.
External links[edit | edit source]
| Classification |
|---|
Categories: [Pathology of pregnancy, childbirth and the puerperium] [Mood disorders] [Psychosis] [Bipolar disorder] [Mental disorders associated with pregnancy, childbirth or the puerperium] [RTT] [WHRTT]
↧ Download as ZWI file | Last modified: 05/13/2024 21:21:12 | 5 views
☰ Source: https://mdwiki.org/wiki/Postpartum_psychosis | License: CC BY-SA 3.0
 KSF
KSF